Topics
Units and Measurements
- Quantitative Science
- System of Units
- Derived Quantities and Units
- Rules and Conventions for Writing SI Units and Their Symbols
- Measurement of Length
- Measurement of Mass
- Measurement of Time
- Dimensions and Dimensional Analysis
- Accuracy, Precision and Uncertainty in Measurement
- Errors in Measurements>Systematic Errors
- Errors in Measurements>Random Errors
- Estimation of Errors
- Combination of Errors
- Significant Figures
- Definitions of SI Units and Constants
Mathematical Methods
- Vector Analysis
- Scalar
- Vector
- Vector Operations>Multiplication of a Vector by a Scalar
- Vector Operations>Addition and Subtraction of Vectors
- Vector Operations>Triangle Law for Vector Addition
- Vector Operations>Law of parallelogram of vectors
- Resolution of Vectors
- Multiplication of Vectors>Scalar Product(Dot Product)
- Multiplication of Vectors>Vector Product (Cross Product)
- Concept of Calculus
- Differential Calculus
- Integral Calculus
Motion in a Plane
- Concept of Motion
- Rectilinear Motion
- Displacement
- Path Length
- Average Velocity
- Average Speed
- Instantaneous Velocity
- Instantaneous Speed
- Acceleration in Linear Motion
- Relative Velocity
- Motion in Two Dimensions - Motion in a Plane
- Average and Instantaneous Velocities
- Acceleration in a Plane
- Equations of Motion in a Plane with Constant Acceleration
- Relative Velocity in Two Dimensions
- Projectile Motion
- Uniform Circular Motion (UCM)
- Key Parameters of Circular Motion
- Centripetal Acceleration
- Conical Pendulum
Laws of Motion
- Fundamental Principles of Motion and Mechanics
- Types of Motion
- Aristotle’s Fallacy
- Newton’s Laws of Motion
- Newton's First Law of Motion
- Newton’s Second Law of Motion
- Newton's Third Law of Motion
- Inertial and Non-inertial Frames of Reference
- Types of Forces>Fundamental Forces in Nature
- Types of Forces>Contact and Non-Contact Forces
- Types of Forces>Real and Pseudo Forces
- Types of Forces>Conservative and Non-Conservative Forces
- Types of Forces>Work Done by a Variable Force
- Work Energy Theorem
- Principle of Conservation of Linear Momentum
- Collisions
- Elastic and Inelastic Collisions
- Perfectly Inelastic Collision
- Coefficient of Restitution e
- Expressions for Final Velocities in Elastic Head-On Collision
- Loss of Kinetic Energy in Perfectly Inelastic Head-On Collision
- Collision in Two Dimensions
- Impulse of a Force
- Necessity of Defining Impulse
- Rotational Analogue of a Force: Moment of a Force Or Torque
- Couple and Its Torque
- Proof of Independence of the Axis of Rotation
- Mechanical Equilibrium
- States of Equilibrium
- Centre of Mass>Mathematical Understanding of Centre of Mass
- Centre of Mass>Velocity of Centre of Mass
- Centre of Mass>Acceleration of Centre of Mass
- Centre of Mass>Characteristics of Centre of Mass
- Centre of Gravity
Gravitation
- Concept of Gravitation
- Kepler’s Laws
- Law of Orbit or Kepler's First Law
- Law of Areas or Kepler's Second Law
- Law of Periods or Kepler's Third Law
- Newton's Universal Law of Gravitation
- Measurement of the Gravitational Constant (G)
- Acceleration Due to Gravity (Earth’s Gravitational Acceleration)
- Variation in the Acceleration>Variation in Gravity with Altitude
- Variation in the Acceleration>Variation in Gravity with Depth
- Variation in the Acceleration>Variation in Gravity with Latitude and Rotation of the Earth
- Variation in the Acceleration>Effect of the Shape of the Earth
- Gravitational Potential Energy
- Expression for Gravitational Potential Energy
- Connection of Potential Energy Formula with mgh
- Potential and Potential Difference
- Escape Velocity
- Earth Satellites
- Projection of Satellite
- Weightlessness in a Satellite
- Time Period of Satellite
- Binding Energy of an Orbiting Satellite
Mechanical Properties of Solids
- Understanding Mechanical Properties of Solids
- Elastic Behavior of Solids
- Stress and Strain
- Types of Stress and Corresponding Strain
- Hooke’s Law
- Elastic Modulus>Young’s Modulus
- Elastic Modulus>Bulk Modulus
- Elastic Modulus>Shear Modulus (Modulus of Rigidity)
- Elastic Modulus>Poisson’s Ratio
- Stress-strain Curve
- Strain Energy
- Hardness of Material
- Friction in Solids
- Origin of Friction
- Types of Friction>Static Friction
- Types of Friction>Kinetic Friction
- Types of Friction>Rolling Friction
Thermal Properties of Matter
- Understanding Thermal Properties of Matter
- Temperature and Heat
- Measurement of Temperature
- Absolute Zero and Absolute Temperature
- Ideal Gas Equation
- Thermal Expansion
- Linear Expansion
- Areal Expansion
- Volume Expansion
- Specific Heat Capacity of Solids and Liquids
- Relation Between Coefficient of Expansion
- Specific Heat Capacity of Gas
- Heat Equation
- Thermal Capacity
- Calorimetry
- Change of State
- Analysis of Observation>From Point A to B
- Analysis of Observation>From Point B to D
- Evaporation vs Boiling
- Boiling Point and Pressure
- Sublimation
- Phase Diagram
- Gas and Vapour
- Latent Heat
- Heat Transfer
- Conduction
- Thermal Conductivity
- Coefficient of Thermal Conductivity
- Thermal Resistance
- Applications of Thermal conductivity
- Convection
- Application of Convection
- Free and Forced Convection
- Radiation
- Newton’s Law of Cooling
Sound
- Sound Waves
- Common Properties of All Waves
- Transverse Waves
- Longitudinal Waves
- Mathematical Expression of a Wave
- The Speed of Travelling Waves
- The Speed of Transverse Waves
- The Speed of Longitudinal Waves
- Newton's Formula for Velocity of Sound
- Laplace’s Correction
- Factors Affecting Speed of Sound
- Principle of Superposition of Waves
- Echo
- Reverberation
- Acoustics
- Qualities of Sound
- Doppler Effect
- Source Moving and Listener Stationary
- Listener Approaching a Stationary Source with Velocity
- Both Source and Listener are Moving
- Common Properties between Doppler Effect of Sound and Light
- Major Differences between Doppler Effects of Sound and Light
Optics
- Fundamental Concepts of Light
- Nature of Light
- Ray Optics Or Geometrical Optics
- Cartesian Sign Convention
- Reflection>Reflection from a Plane Surface
- Reflection>Reflection from Curved Mirrors
- Total Internal Reflection
- Refraction of Light
- Applications of Total Internal Reflection
- Refraction at a Spherical Surface and Lenses
- Thin Lenses and Their Combination
- Refraction at a Single Spherical Surface
- Lens Makers' Equation
- Dispersion of Light
- Analysis of Prism
- Thin Prisms
- Some Natural Phenomena Due to Sunlight
- Defects of Lenses
- Optical Instruments
- Simple Microscope or a Reading Glass
- Compound Microscope
- Telescope
Electrostatics
- Concept of Electrostatics
- Electric Charge
- Basic Properties of Electric Charge
- Additive Nature of Charge
- Quantization of Charge
- Conservation of Charge
- Force between Charges
- Coulomb’s Law
- Scalar Form of Coulomb’s Law
- Relative Permittivity or Dielectric Constant
- Definition of Unit Charge from the Coulomb’s Law
- Coulomb's Law in Vector Form
- Principle of Superposition
- Electric Field
- Electric Field Intensity Due to a Point-Charge
- Practical Way of Calculating Electric Field
- Electric Lines of Force
- Electric Flux
- Gauss’s Law
- Electric Dipole
- Couple Acting on an Electric Dipole in a Uniform Electric Field
- Electric Intensity at a Point Due to an Electric Dipole
- Continuous Charge Distribution
Electric Current Through Conductors
- Electric Current and Its Related Concepts
- Electric Current
- Flow of Current Through a Conductor
- Drift Speed
- Ohm's Law
- Limitations of Ohm’s Law
- Electrical Power
- Resistors
- Rheostat
- Resistors in Parallel
- Specific Resistance or Electrical Resistivity
- Variation of Resistance with Temperature
- Electromotive Force (emf)
- Cells in Series
- Cells in Parallel
- Types of Cells
Magnetism
- Concept of Magnetism
- Magnetic Lines of Force
- Bar Magnet and Solenoid Analogy
- Magnetic Field due to a Bar Magnet
- Magnetic Field Due to a Bar Magnet at an Arbitrary Point
- Gauss' Law of Magnetism
- The Earth’s Magnetism
Electromagnetic Waves and Communication System
- Foundations of Electromagnetic Theory
- Concept of Electromagnetic Waves
- Concept of Electromagnetic Waves
- Characteristics of EM Waves
- Electromagnetic Spectrum
- Radio Waves
- Microwaves
- Infrared waves
- Visible Light
- Ultraviolet rays
- X-rays
- Gamma Rays
- Propagation of EM Waves
- Ground (surface) Wave
- Space wave
- Sky wave propagation
- Communication System
- Elements of a Communication System
- Commonly Used Terms in Electronic Communication System
- Modulation
Semiconductors
- Concept of Semiconductors
- Electrical Conduction in Solids
- Band Theory of Solids
- Intrinsic Semiconductor
- Extrinsic Semiconductor
- n-type Semiconductor
- p-type Semiconductor
- Charge neutrality of extrinsic semiconductors
- Diode or p-n Junction
- Basics of Semiconductor Devices
- Applications of Semiconductors and P-n Junction Diode
- Thermistor
- Introduction
- Definition: Absolute Zero
- Definition: Kelvin Scale
- Definition: Triple Point
- Definition: Ideal Gas
- Definition: Universal Gas Constant (R)
- Definition: Extrapolation
- Definition: Kelvin
- Definition: Absolute Temperature
- Conversion Formulas
- Gases Respond to Temperature
- The Discovery of Absolute Zero
- The Kelvin (Absolute) Temperature Scale
- Temperature Scales & Conversion
- Law: Boyle's Law
- Law: Charles' Law
- Law: Gay-Lussac's Law
- The Ideal Gas Equation
- Example 1
- Example 2
- Key Points: Absolute Zero and Absolute Temperature
Introduction
Every gas thermometer in a hospital, every weather balloon, and every rocket engine relies on how gas pressure, volume, and temperature are related. This topic lays the foundation for thermodynamics — and it all starts with defining temperature properly.
Definition: Absolute Zero
The lowest theoretically possible temperature (0 K = −273.15 °C), where ideal gas molecules have zero kinetic energy.
OR
The lowest attainable temperature, obtained by plotting the relation between pressure of the gas vs its temperature, where all lines for different gases cut the temperature axis at the same point (−273.15°C), is called the absolute zero of temperature.
Definition: Kelvin Scale
The SI absolute temperature scale starting at absolute zero. Written as K (no degree symbol °).
Definition: Triple Point
The unique temperature & pressure at which solid, liquid, and gas phases of a substance coexist in equilibrium.
OR
The temperature where the solid, liquid, and gas state of a material co-exist in equilibrium, and this occurs only at a unique temperature and pressure, is called the triple point.
Definition: Ideal Gas
A hypothetical gas whose molecules have no volume and exert no intermolecular forces; obeys PV = μRT exactly.
Definition: Universal Gas Constant (R)
A constant in the ideal gas equation; R = 8.31 J mol⁻¹ K⁻¹.
Definition: Extrapolation
Extending a graph line beyond the measured data to predict values.
Definition: Kelvin
One kelvin = 1/273.16 of the difference between absolute zero and the triple point of water.
Definition: Absolute Temperature
The temperature scale where −273.15°C corresponds to 0 K, i.e., the temperature at which the pressure of a gas would become zero, is called the absolute temperature (0 K).
Conversion Formulas
Master Conversion Formula:
\[\frac {T_C}{100}\] = \[\frac {(T_{F}-32)}{180}\] = \[\frac {(T_{K}-273.15)}{100}\]
Gases Respond to Temperature
Experiments with gases at low densities reveal a simple and powerful pattern:
- At constant pressure: The volume of a gas is directly proportional to its temperature (°C). → Charles' Law
- At constant volume: The pressure of a gas is directly proportional to its temperature (°C). → Gay-Lussac's Law
V ∝ TC (at constant P)
P ∝ TC (at constant V)
The Graphs Show:
When we plot these relationships, we get straight lines — but with important properties:
- The lines do not pass through the origin (non-zero y-intercept).
- Different gases have different slopes.
- Gases expand linearly with temperature: equal temperature rises cause equal increases in volume (or pressure).
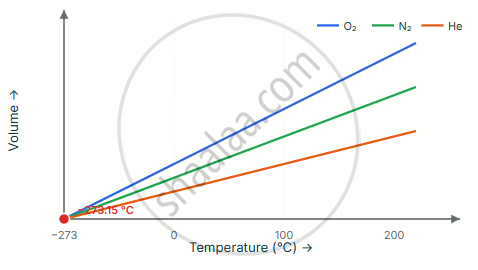
Fig: Volume vs Temperature at Constant Pressure
The Discovery of Absolute Zero
If we extrapolate the P–T graph backwards — imagining the gas doesn't liquefy — we ask: "At what temperature would the gas pressure drop to zero?"
The answer, from extrapolation, is:
This is called absolute zero.
Why This Is Remarkable:
- All gases, regardless of type, produce lines that converge to the same point: −273.15 °C.
- This point does not depend on which specific gas is used — it is a universal property of nature.
- No temperature lower than absolute zero is physically possible.
- In practice, absolute zero has never been achieved — only approached.
The Kelvin (Absolute) Temperature Scale
The old two-fixed-point system (ice point + steam point) had a practical problem: the boiling point of water is very sensitive to pressure changes, making calibration difficult. In 1954, scientists adopted a one-fixed-point scale based on the triple point of water.
The triple point is the unique combination of temperature and pressure at which all three phases of a substance — solid, liquid, and gas — coexist in perfect equilibrium.
| Property | Value |
|---|---|
| Triple point temperature | 273.16 K (= 0.01 °C) |
| Triple point pressure | 6.11 × 10⁻³ atm (= 611 Pa) |
The Kelvin
- The size of one kelvin is identical to one degree Celsius.
- Kelvin is written as K (no degree symbol °). Named after Lord Kelvin (1824–1907).
TK = TC + 273.15
Common Mistake:
Students often write "°K" — this is incorrect. The correct notation is simply K (e.g., 300 K, not 300°K).Why Is It Called "Absolute"?
- Based on a universal property of ideal gases, not on any particular substance.
- Its zero point represents the lowest physically possible temperature.
- Unlike the Celsius or Fahrenheit scales, the Kelvin scale has no negative values.
Temperature Scales & Conversion
Scale Comparison Table
| Fixed Point | Kelvin (K) | Celsius (°C) | Fahrenheit (°F) |
|---|---|---|---|
| Absolute zero | 0 | −273.15 | −459.67 |
| Triple point of water | 273.16 | 0.01 | 32.018 |
| Ice point (melting) | 273.15 | 0 | 32 |
| Steam point (boiling) | 373.15 | 100 | 212 |
| Human body temperature | ~310 | ~37 | ~98.6 |
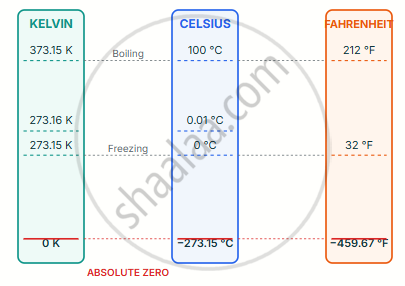
Three Temperature Scales — Side by Side
"Celsius Adds 273" — To go from Celsius to Kelvin, always add 273.15. To go the other way, subtract. The interval sizes are identical: a change of 1 °C = a change of 1 K.
Law: Boyle's Law
When temperature is constant, the product of pressure and volume of a gas remains constant.
Law: Charles' Law
When pressure is constant, the ratio of volume to temperature of a gas remains constant.
Law: Gay-Lussac's Law
When volume is constant, the ratio of pressure to temperature of a gas remains constant.
The Ideal Gas Equation
Building the Equation Step by Step
The ideal gas equation combines two experimental gas laws:
| Law | Statement | Mathematical Form |
|---|---|---|
| Boyle's Law | At constant T, P and V are inversely proportional. | PV = constant |
| Charles' Law | At constant P, V is directly proportional to T. | V/T = constant |
Combining both laws:
\[\frac {PV}{T}\] = constant
For μ moles of any ideal gas, this constant equals μR, giving us:
PV = μRT
| Symbol | Quantity | SI Unit |
|---|---|---|
| P | Pressure | Pascal (Pa) |
| V | Volume | m³ |
| μ | Number of moles | mol |
| R | Universal gas constant | 8.31 J mol⁻¹ K⁻¹ |
| T | Absolute temperature | Kelvin (K) only |
The ideal gas equation only works with Kelvin. If the problem gives temperature in Celsius, you must convert to Kelvin first: T(K) = T(°C) + 273.15. Using °C directly will yield an incorrect answer.
Example 1
= \[\frac {9}{5}\](24.57 − 273.15) + 32
= \[\frac {9}{5}\](−248.58) + 32
= −447.44 + 32 = −415.44 °F
Example 2
100y − 3200 = 180y − 49167
80y = 45967
y = 574.59
Key Points: Absolute Zero and Absolute Temperature
- Gases expand linearly with temperature, making them useful for thermometers. This consistent behaviour suggests the existence of a lowest temperature limit.
- Absolute zero (−273.15 °C or 0 K) is the temperature where an ideal gas would have zero pressure. It is the lowest possible temperature.
- The Kelvin scale begins at absolute zero and uses the triple point of water (273.16 K) as a reference point. It is the SI temperature scale.
- The ideal gas equation (PV = μRT) combines all gas laws into a single relationship among pressure, volume, and temperature. It works best for gases at low pressure and high temperature.
