Topics
Force
Force, Work, Power and Energy
Light
Work, Energy and Power
- Concept of Work
- Measurement of Work
- Expression for Work (W = F S cosθ)
- Work Done by the Force of Gravity (W = mgh)
- Concept of Power
- Work vs Power
- Concept of Energy
- Energy vs Power
- Mechanical Energy > Potential Energy (U)
- Mechanical Energy > Kinetic Energy (K)
- Potential vs Kinetic Energy
- Conversion of Potential Energy into Kinetic Energy
- Forms of Energy > Solar Energy
- Forms of Energy > Heat Energy
- Forms of Energy > Light Energy
- Forms of Energy > Chemical Energy
- Forms of Energy > Hydro Energy
- Forms of Energy > Electrical Energy
- Forms of Energy > Nuclear Energy
- Forms of Energy > Geo Thermal Energy
- Forms of Energy > Wind Energy
- Forms of Energy > Sound Energy
- Forms of Energy > Magnetic Energy
- Forms of Energy > Mechanical Energy
- Conversion of Energies
- Principle of Conservation of Energy
- Proof: Kinetic + Potential Energy = Constant for Free Fall
- Application of the Principle of Conservation of Energy
Sound
Machines
- Concept of Machines
- Technical Terms Related to a Machine
- Principle of a Machine
- Efficiency, Mechanical Advantage, and Velocity Ratio
- Levers
- Types of Levers
- Pulley
- Single Fixed Pulley
- A Single Movable Pulley
- Single Pulley vs Single Movable Pulley
- Combination of Pulleys
- Using one fixed pulley and other movable pulleys
- Using several pulleys in two blocks (block and tackle system)
Electricity and Magnetism
Refraction of Light at Plane Surfaces
- Light: Reflection and Refraction
- Refraction of Light
- Laws of Refraction
- Speed of Light in Different Media
- Principle of Reversibility of the Path of Light
- Refraction Laws & Glass Index
- Refraction of Light Through a Rectangular Glass Block
- Multiple Images in a Thick Mirror
- Prism
- Refraction of Light Through a Prism
- Real and Apparent Depth
- Apparent Bending of a Stick Under Water
- Consequences of Refraction of Light
- Transmission of Light From a Denser Medium to a Rarer Medium
- Critical Angle
- Relationship between Critical Angle and Refractive Index
- Total Internal Reflection
- Total Internal Reflection in a Prism
- Total Internal Reflection Through a Right-Angled Isosceles Prism
- Total Internal Reflection Through an Equilateral Prism
- Total Internal Reflection Through Right-angled prism
- Use of a Total Internal Reflecting Prism in Place of a Plane Mirror
- Total Internal Reflection vs Reflecting from a Plane Mirror
- Consequences of Total Internal Refraction
Refraction Through a Lens
- Concept of Lenses
- Action of a Lens as a Set of Prisms
- Technical Terms Related to a Lens
- Convex Lens vs Concave Lens
- Refraction of Light Through an Equi-Convex Lens and an Equi-Concave Lens
- Principal Rays for Ray Diagrams
- Real Image vs Virtual Image
- Construction of a Ray Diagram for a Lens
- Images Formed by Convex Lenses
- Images Formed by Concave Lenses
- Sign Convention
- Lens Formula
- Linear Magnification
- Power of a Lens
- Magnifying Glass Or Simple Microscope
- Application of Lenses
- Experimental Determination of Focal Length of Convex Lens
- Convex Lens vs Concave Lens
Heat
Spectrum
- Deviation Produced by a Triangular Prism
- Colour in White Light with Their Wavelength and Frequency Range
- Dispersion of Light
- Electromagnetic Spectrum
- Properties and Uses of Different Radiations of the Electromagnetic Spectrum
- Distinction between Ultraviolet, Visible, and Infrared Radiations
- Scattering of Light
- Applications of Scattering of Light
Modern Physics
Sound
- Sound Waves
- Light Waves vs Sound Waves
- Reflection of Sound Waves
- Echo
- Determination of Speed of Sound by the Method of Echo
- Use of Echoes
- Natural Vibrations
- Damped Vibrations
- Natural Vibrations vs Damped Vibrations
- Forced Vibrations
- Natural Vibrations vs Forced Vibrations
- Resonance (a special case of forced vibrations)
- Demonstration of Resonance
- Forced Vibrations vs Resonant Vibrations
- Examples of Resonance
- Characteristics of Sound
- Loudness and Intensity
- Pitch and frequency
- Quality and Wave Form
- Music and Noise
Current Electricity
- Electric Charge
- Electric Current
- Potential and Potential Difference
- Resistance
- Ohm's Law
- Experimental Verification of Ohm’s Law and Ohmic Resistors
- Ohmic and Non-ohmic Resistors
- Specific Resistance or Electrical Resistivity
- Choice of Material of a Wire
- Superconductors
- Electro-Motive Force of a Cell
- Terminal Voltage of a Cell
- Internal Resistance of a Cell
- Resistance of a System of Resistors
- Resistors in Series
- Resistors in Parallel
- Forms of Energy > Electrical Energy
- Measurement of Electrical Energy
- Electrical Power
- Commercial Unit of Electrical Energy
- Power Rating of Common Electrical Appliances
- Household Consumption of Electric Energy
- Heating Effect of Electric Current
Household Circuits
- Transmission of Power from the Power Generating Station to the Consumer
- Power Distribution to a House
- House Wiring (Ring System)
- Fuse
- Reason for connecting the fuse in the live wire
- Current Rating of a Fuse
- Switches
- Circuits with Dual Control Switches (Staircase Wire)
- Earthing
- Three-pin Plug and Socket
- Colour Coding of Wires in a Cable
- High Tension Wires
- Precautions to Be Taken While Using Electricity
Electro-Magnetism
- Oersted's Experiment
- Applications of Biot-Savart's Law > Magnetic Field due to a Finite Straight Current-Carrying Wire
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Electromagnet
- Permanent Magnet
- Comparison of an Electro Magnet with a Permanent Magnet
- Advantages of an Electromagnet over a Permanent Magnet
- Uses of Electromagnet
- Force on a Current Carrying Conductor in a Magnetic Field
- Simple D.C. Motor
- Electromagnetic Induction
- Demonstration of the Phenomenon of Electromagnetic Induction
- Faraday's Explanation
- Faraday's Laws of Electromagnetic Induction
- A.C. Generator
- Frequency of an a.c. in Household Supplies
- Comparison Between A.C. Generator and D.C. Motor
- Transformers
Calorimetry
- Heat
- The Temperature and a Thermometer
- Factors Affecting the Quantity of Heat Absorbed to Increase the Temperature of a Body
- Heat vs Temperature
- Thermal or Heat Capacity
- Specific Heat Capacity
- Heat Capacity vs Specific Heat Capacity
- Specific Heat Capacity of Some Common Substances
- Calorimetry
- Principle of Method of Mixtures or Principle of Calorimetry
- Natural Phenomena and Consequences of High Specific Heat Capacity of Water
- Examples of High and Low Heat Capacity
- Change of State
- Melting and Freezing
- Melting Point and Its Effects
- Vaporisation or Boiling
- Boiling Point and Its Effects
- Latent Heat
- Specific Latent Heat of Fusion of Ice
- Explanation of Latent Heat of Melting based on Kinetic Model
- Natural Consequences of High Specific Latent Heat of Fusion of Ice
Radioactivity
- Structure of the Atom and Nucleus
- Atomic Models
- Isotopes
- Isobars
- Isotones
- Radioactivity
- Radioactivity as emission of Alpha, Beta, and Gamma Radiations
- Properties of Alpha Particles
- Properties of Beta Particles
- Properties of Gamma Radiations
- Distinction between the Properties of α, β, and γ Radiations
- Changes Within the Nucleus in Alpha, Beta and Gamma Emission
- Uses of Radioactive Isotopes
- Sources of Harmful Radiations
- Harmful Effects of Radiation
- Safety Precautions While Using Nuclear Energy
- Background Radiations
- Forms of Energy > Nuclear Energy
- Nuclear Fission
- Radioactive Decay Vs Nuclear Fission
- Nuclear Fusion
- Difference between Nuclear Fusion and Neclear Fission
- Introduction
- Definition: Calorimetry
- Definition: Calorimeter
- Calorimeter
- Law: Principle of Calorimetry
- Experiment: Method of Mixtures
- Derivation of the Calorimetry Equation
- Example
- Key Points: Calorimetry
Introduction
A thermometer tells you how hot something is, but it doesn't tell you how much heat energy an object contains or exchanges. To answer that question, scientists developed a technique called calorimetry and a device called a calorimeter.
Definition: Calorimetry
Calorimetry is the science of measuring heat exchange during physical or chemical processes. The word comes from the Latin calor (heat) + Greek metron (measure).
OR
An experimental technique for the quantitative measurement of heat exchange is called calorimetry.
CISCE: Class 10
Definition: Calorimeter
A calorimeter is a cylindrical vessel which is used to measure the amount of heat gained (or lost) by a body when it is mixed with another body or substance.
Calorimeter
A calorimeter is an insulated device used to measure the heat gained or lost by a substance during a thermal process. It works on a simple idea: trap all the heat inside the device so nothing escapes, and then measure the temperature change.
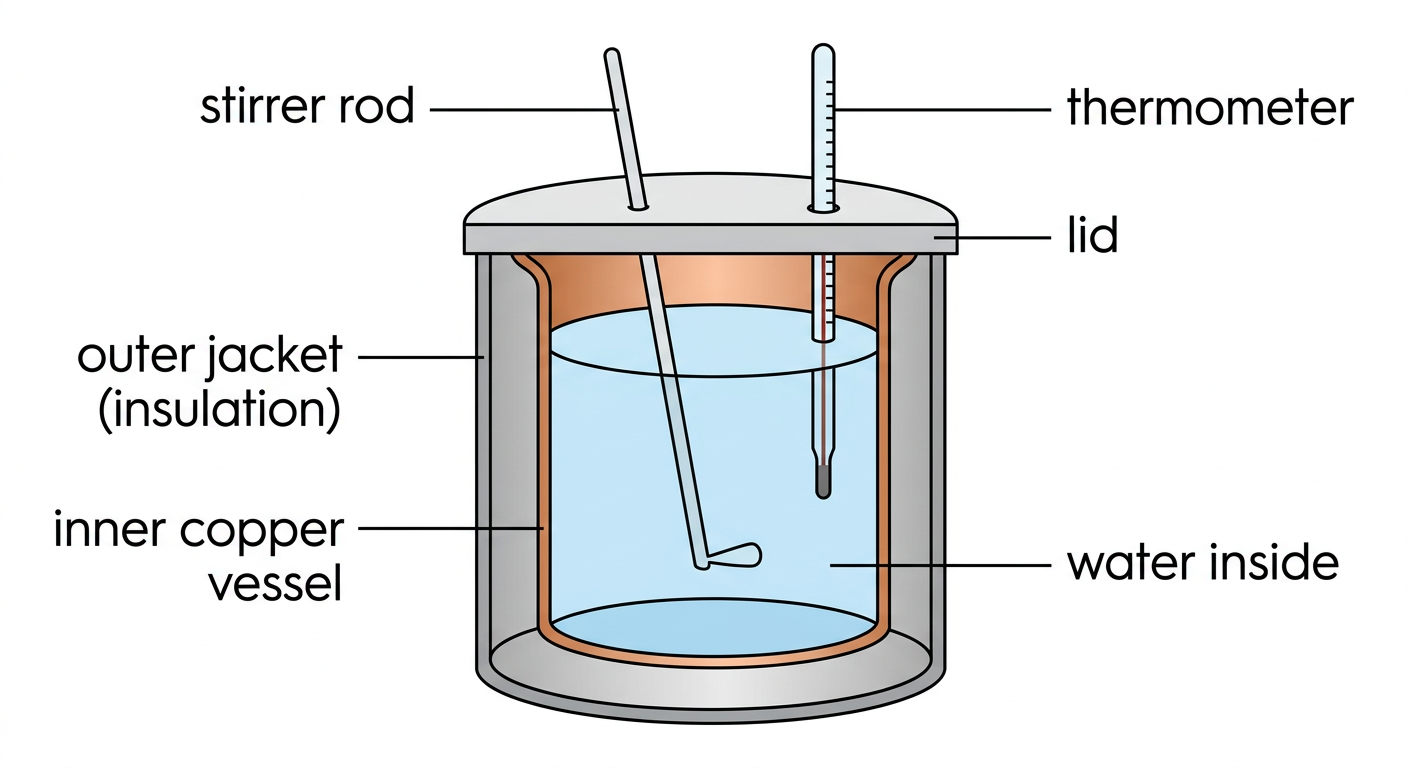
Fig.: Cross-section of a simple water calorimeter showing all key components
| Part | Material | Function |
|---|---|---|
| Inner Vessel | Copper or Aluminium | Holds water; copper is an excellent thermal conductor for quick heat exchange |
| Outer Jacket | Insulated material | Prevents heat loss to the surroundings |
| Insulating Air Gap | Air / Padding | Creates thermal isolation between inner and outer vessels |
| Stirrer | Metal (copper) | Ensures uniform temperature distribution throughout the water |
| Thermometer | Glass / Digital | Measures the temperature of the water accurately |
| Lid | Insulated | Prevents heat escape from the top |
Fig.: A calorimeter works just like a thermos flask — both trap heat inside using insulation
Law: Principle of Calorimetry
Statement: When different parts of an isolated system are at different temperatures, heat transfers from the part at higher temperature to the part at lower temperature. The heat lost by the hot object is equal to the heat gained by the cold object, provided no heat is allowed to escape to the surroundings.
(For liquid in calorimeter: m1c1Δθ + mcccΔθ)
Key Points:
- A system is said to be isolated if no exchange of heat occurs between the system and its surroundings.
- Calorimetry literally means measurement of heat.
- Energy supplied by heater = VIt (voltage × current × time).
- This principle is based on the Law of Conservation of Energy.
Experiment: Method of Mixtures
The method of mixtures is the standard experimental technique for determining the specific heat capacity of an unknown substance using a calorimeter.
Procedure:
- Fill the Calorimeter: Pour a known mass of water (m₃) into the inner copper vessel. Note the initial temperature (T₂) of both water and calorimeter.
- Heat the Sample: Heat the sample (substance whose specific heat you want to find) to a high temperature (T₁). For example, place it in boiling water at 100°C.
- Quick Transfer: Quickly transfer the hot sample into the calorimeter water. Speed matters — minimise heat loss during the transfer!
- Stir Continuously: Use the stirrer to ensure uniform temperature distribution throughout the water.
- Record Final Temperature: Wait until the thermometer shows a stable reading. This is the final equilibrium temperature (T).
- Apply the Formula: Use the calorimetry equation: Heat lost by sample = Heat gained by water + Heat gained by calorimeter.
Derivation of Calorimetry Equation
Let's build the master formula step by step. First, define all our variables clearly.
Variable Definitions
| Symbol | Meaning |
|---|---|
| m₁ | Mass of the hot sample |
| m₂ | Mass of the calorimeter (+ stirrer) |
| m₃ | Mass of water in the calorimeter |
| s₁ | Specific heat capacity of the sample (unknown) |
| s₂ | Specific heat capacity of calorimeter material (e.g., copper = 387 J kg⁻¹ K⁻¹) |
| s₃ | Specific heat capacity of water = 4186 J kg⁻¹ K⁻¹ |
| T₁ | Initial temperature of the hot sample |
| T₂ | Initial temperature of calorimeter + water |
| T | Final equilibrium temperature of the entire system |
Step-by-Step Derivation
The sample cools from T₁ down to T, so ΔT = T₁ − T
The calorimeter heats up from T₂ to T, so ΔT = T − T₂
Water also heats from T₂ to T (same ΔT as the calorimeter)
m₁ s₁ (T₁ − T) = m₂ s₂ (T − T₂) + m₃ s₃ (T − T₂) ... Eq. (1)
This is the master formula. If you know all masses, temperatures, and specific heats of water and copper, you can calculate s₁ for any unknown material.
Example
Problem: A 0.06 kg aluminium sphere is heated to 100°C and placed in a 0.12 kg copper calorimeter containing 0.30 kg of water at 25°C. The mixture reaches a steady temperature of 28°C. Find the specific heat capacity of aluminium.
Solution:
Given:
Mass of aluminium sphere (m₁) = 0.06 kg
Mass of copper calorimeter (m₂) = 0.12 kg
Mass of water (m₃) = 0.30 kg
Specific heat of copper (s₂) = 387 J kg⁻¹ K⁻¹
Specific heat of water (s₃) = 4180 J kg⁻¹ K⁻¹
Initial temp of sphere (T₁) = 100°C
Initial temp of water + calorimeter (T₂) = 25°C
Final equilibrium temp (T) = 28°C
Step 1: Calculate temperature changes
- Temperature drop of sphere: T₁ − T = 100 − 28 = 72°C
- Temperature rise of water & calorimeter: T − T₂ = 28 − 25 = 3°C
Step 2: Apply Eq.
Step 3: Substitute values
Step 4: Compute the numerator
- 0.12 × 387 = 46.44
- 0.30 × 4180 = 1254
- Sum = 46.44 + 1254 = 1300.44
- Numerator = 1300.44 × 3 = 3901.32
Step 5: Compute the denominator
- 0.06 × 72 = 4.32
Step 6: Final answer
Key Points: Calorimetry
- A calorimeter is an insulated device used to measure heat transfer; measurement of specific heat of a substance is carried out using it.
- Principle of Calorimetry: Heat lost by hot body = Heat gained by cold body, which represents the law of conservation of heat energy.
- In the method of mixtures, a heated sample is placed in the calorimeter and the temperature change is measured to calculate specific heat using the formula Q = msΔt.
- Specific heat of a substance depends on the nature of the substance; water is preferred in calorimetry due to its high specific heat, allowing it to absorb large amounts of heat with minimal temperature change.
- For accurate results, the sample must be transferred quickly into the calorimeter and stirred well to ensure uniform heat distribution.

