Advertisements
Advertisements
प्रश्न
State the order of filling atomic orbitals following Aufbau principle.
Advertisements
उत्तर
Aufbau principle:
- The Aufbau principle gives the sequence in which various orbitals are filled with electrons.
- In the ground state of an atom, the orbitals are filled with electrons based on the increasing order of energies of orbitals, Pauli’s exclusion principle, and Hund’s rule of maximum multiplicity.
- Increasing order of energies of orbitals:
a. Orbitals are filled in order of increasing value of (n + 1)
b. In cases where the two orbitals have the same value of (n + 1), the orbital with a lower value of n is filled first. - The increasing order of energy of different orbitals in a multi-electron atom is:
1s < 2s < 2p < 3s < 3p < 4s < 3d < 4p < 5s < 4d < 5p < 6s < 4f < 5d < 6p < 7s and so on.
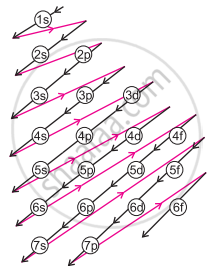
Increasing order of orbital energy
APPEARS IN
संबंधित प्रश्न
Using s, p, d notations, describe the orbital with the following quantum numbers n = 4; l = 2.
Choose the correct option.
Principal Quantum number describes -
Give the names of quantum numbers.
Define the term Electronic configuration.
State Hund’s rule of maximum multiplicity with a suitable example.
Explain the anomalous behaviour of copper.
Write orbital notations for the electron in orbitals with the following quantum numbers.
n = 2, l = 1
Write orbital notations for the electron in orbitals with the following quantum numbers.
n = 3, l = 2
Write condensed orbital notation of electronic configuration of the following element:
Carbon (Z = 6)
Write condensed orbital notation of electronic configuration of the following element:
Oxygen (Z = 8)
Write condensed orbital notation of electronic configuration of the following element:
Silicon (Z = 14)
Draw shapes of 2s orbitals.
The electronic configuration of oxygen is written as 1s2 2s2 \[\ce{2p^2_{{x}}}\] \[\ce{2p^1_{{y}}}\] \[\ce{2p^1_{{z}}}\] and not as 1s2 2s2 \[\ce{2p^2_{{x}}}\], \[\ce{2p^2_{{y}}}\] \[\ce{2p^0_{{z}}}\], Explain.
Write a note on ‘Principal Quantum number.
Indicate the number of unpaired electrons in \[\ce{Si}\] (Z = 14).
Which mineral among the following contains vanadium in it?
The designation of a subshell with n = 6 and l = 2 is ____________.
How many electrons can fit in the orbital for which n = 4 and l = 2?
Which of the following options does not represent ground state electronic configuration of an atom?
Out of the following pairs of electrons, identify the pairs of electrons present in degenerate orbitals:
| (i) | (a) `n = 3, l = 2, m_l = -2, m_s = - 1/2` |
| (b) `n = 3, l = 2, m_l = -1, m_s = - 1/2` | |
| (ii) | (a) `n = 3, l = 1, m_l = 1, m_s = + 1/2` |
| (b) `n = 3, l = 2, m_l = 1, m_s = + 1/2` | |
| (iii) | (a) `n = 4, l = 1, m_l = 1, m_s = + 1/2` |
| (b) `n = 3, l = 2, m_l = 1, m_s = + 1/2` | |
| (iv) | (a) `n = 3, l = 2, m_l = +2, m_s = - 1/2` |
| (b) `n = 3, l = 2, m_l = +2, m_s = + 1/2` |
Calculate the total number of angular nodes and radial nodes present in 3p orbital.
The arrangement of orbitals on the basis of energy is based upon their (n + l) value. Lower the value of (n + l), lower is the energy. For orbitals having same values of (n + l), the orbital with lower value of n will have lower energy.
Based upon the above information, solve the questions given below:
Which of the following orbitals has the lowest energy?
5p, 5d, 5f, 6s, 6p
What is the difference between the terms orbit and orbital?
Choose the INCORRECT statement
Which one of the following laws will represent the pairing of electrons in a subshell after each orbital is filled with one electron?
