Advertisements
Advertisements
Question
State the order of filling atomic orbitals following Aufbau principle.
Advertisements
Solution
Aufbau principle:
- The Aufbau principle gives the sequence in which various orbitals are filled with electrons.
- In the ground state of an atom, the orbitals are filled with electrons based on the increasing order of energies of orbitals, Pauli’s exclusion principle, and Hund’s rule of maximum multiplicity.
- Increasing order of energies of orbitals:
a. Orbitals are filled in order of increasing value of (n + 1)
b. In cases where the two orbitals have the same value of (n + 1), the orbital with a lower value of n is filled first. - The increasing order of energy of different orbitals in a multi-electron atom is:
1s < 2s < 2p < 3s < 3p < 4s < 3d < 4p < 5s < 4d < 5p < 6s < 4f < 5d < 6p < 7s and so on.
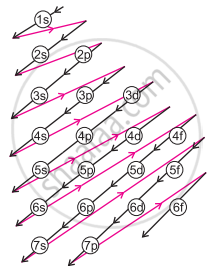
Increasing order of orbital energy
APPEARS IN
RELATED QUESTIONS
State Heisenberg uncertainty principle.
Define the term Electronic configuration.
State and explain Pauli’s exclusion principle.
State Hund’s rule of maximum multiplicity with a suitable example.
Explain the anomalous behaviour of copper.
Explain the anomalous behaviour of chromium.
Write orbital notations for the electron in orbitals with the following quantum numbers.
n = 4, l = 2
Write condensed orbital notation of electronic configuration of the following element:
Lithium (Z = 3)
Write condensed orbital notation of electronic configuration of the following element:
Chlorine (Z = 17)
Draw shapes of 2s orbitals.
If n = 3, what are the quantum number l and m?
Using the concept of quantum numbers, calculate the maximum numbers of electrons present in the ‘M’ shell. Give their distribution in shells, subshells, and orbitals.
How many electrons can fit in the orbital for which n = 4 and l = 2?
Which of the following has a greater number of electrons than neutrons?
(Mass number of Mg, C, O and Na is 24, 12, 16 and 23 respectively).
Which one of the following is NOT possible?
Which of the following properties of atom could be explained correctly by Thomson Model of atom?
Number of angular nodes for 4d orbital is ______.
The arrangement of orbitals on the basis of energy is based upon their (n + l) value. Lower the value of (n + l), lower is the energy. For orbitals having same values of (n + l), the orbital with lower value of n will have lower energy.
Based upon the above information, arrange the following orbitals in the increasing order of energy.
5p, 4d, 5d, 4f, 6s
The arrangement of orbitals on the basis of energy is based upon their (n + l) value. Lower the value of (n + l), lower is the energy. For orbitals having same values of (n + l), the orbital with lower value of n will have lower energy.
Based upon the above information, solve the questions given below:
Which of the following orbitals has the lowest energy?
5p, 5d, 5f, 6s, 6p
Match species given in Column I with the electronic configuration given in Column II.
| Column I | Column II |
| (i) \[\ce{Cr}\] | (a) [Ar]3d84s0 |
| (ii) \[\ce{Fe^{2+}}\] | (b) [Ar]3d104s1 |
| (iii) \[\ce{Ni^{2+}}\] | (c) [Ar]3d64s0 |
| (iv) \[\ce{Cu}\] | (d) [Ar] 3d54s1 |
| (e) [Ar]3d64s2 |
Choose the INCORRECT statement
In assigning R - S configuration, which among the following groups has highest priority?
Which of the following element do not follow Aufbau principle?
Which one of the following laws will represent the pairing of electrons in a subshell after each orbital is filled with one electron?
