Definitions [3]
Primary amines (1°) are the ammonia derivatives in which one H-atom has been replaced by one alkyl or aryl group.
Tertiary amines (3°) are the ammonia derivatives in which three hydrogen atoms have been replaced by three alkyl or aryl group.
Secondary amines (2°) are the ammonia derivatives in which two hydrogen atoms have been replaced by two alkyl or aryl groups.
Key Points
- Amines are derivatives of ammonia (NH₃) where one, two, or all three hydrogen atoms are replaced by alkyl or aryl groups.
- They contain a nitrogen atom with a lone pair of electrons → this lone pair makes amines basic in nature.
- Compounds of nitrogen connected to a carbonyl group are called amides (R–CO–NR'R'') — different from amines.
- Amines overview: Amines are derivatives of ammonia (NH₃) where one or more H-atoms are replaced by alkyl/aryl groups; they can be saturated or unsaturated.
- Classification by source: Aliphatic amines (e.g., CH₃–NH₂, methylamine) and Aromatic amines (e.g., C₆H₅–NH₂, aniline).
- Classification by H-substitution: Primary (1°, –NH₂, e.g., methylamine), Secondary (2°, –NH–, e.g., dimethylamine), and Tertiary (3°, –N–, e.g., trimethylamine).
- Sub-types of 2° and 3° amines: Simple/symmetrical (same groups, e.g., (C₂H₅)₂NH, (C₂H₅)₃N) and Mixed/unsymmetrical (different groups, e.g., C₂H₅–NH–CH₃, C₂H₅–N(CH₃)₂).
- Examples of each class: 1° – n-butylamine, sec-butylamine, tert-butylamine, aniline; 2° – dimethylamine, diethylamine, diphenylamine; 3° – trimethylamine, triethylamine, triphenylamine.
In amines, N atom is sp³ hybridised. Among four sp³ hybridised orbital one contains lone pair of electrons and other three contain bond pairs of electrons.
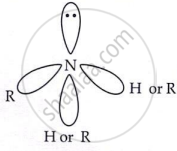
Due to a lone pair of electrons, it is pyramidal in shape.
- Boiling point vs non-polar compounds: Due to intermolecular H-bonding, amines boil at a higher temperature than non-polar compounds such as hydrocarbons.
- Boiling point vs alcohols/acids: The boiling points of amines are less than those of alcohols and carboxylic acids.
- Solubility of lower amines: Lower members of aliphatic amines are soluble in water due to intermolecular H-bonding with water.
- Effect of alkyl group size: Solubility in water decreases as the size of the alkyl group increases.
- Order of boiling points & aromatic amines: Propane < ethylamine < diethylamine < n-butylamine < ethyl alcohol < propanoic acid; aromatic amines are insoluble in water due to the bulky size of the phenyl group.
- Lower aliphatic amines: Used as solvents.
- Aromatic amines: Used in the manufacture of polymers, dyes (azo dyes), and in the rubber industry.
- Quaternary ammonium salts: Used as detergents (e.g., cetyltrimethylammonium bromide).
- Aniline: Starting material for dyes (e.g., aniline dyes), drugs (sulphonamide drugs), and polymers.
| S. No. | Test | 1° amine | 2° amine | 3° amine |
|---|---|---|---|---|
| 1. | Reaction with nitrous acid | Give alcohol with effervescence of N₂ gas | Gives oily nitroso-amine which gives Liebermann’s nitroso amine test | In the cold, it forms nitrite, which is soluble in water and, upon heating, yields nitrosoamine |
| 2. | Hinsberg’s reagent | Gives N-alkyl benzene sulphonamide soluble in alkali | Gives N, N-dialkyl benzene sulphonamide insoluble in alkali | No reaction |
| 3. | Carbylamine test | Forms isocyanide with pungent odour | No reaction | No reaction |
| 4. | Hofmann’s mustard oil reaction | Forms isothio cyanate having smell of mustard oil | No reaction | No reaction |
Only 1° amines (aliphatic and aromatic) react with CHCl₃ and alc. KOH to give isocyanides (carbylamines) with very bad smell.
- Used as a test for 1° amines (Carbylamine test / Isocyanide test).
- 2° and 3° amines do NOT give this reaction.
\[ \underset{1^\circ \text{ amine}}{\mathrm{RNH}_2} + \mathrm{CHCl}_3 + \text{Alc. } 3\mathrm{KOH} \longrightarrow \underset{\text{Alkyl isocyanide}}{\mathrm{RNC}} + 3\mathrm{KCl} + 3\mathrm{H}_2\mathrm{O} \]
HNO₂ is generated in situ: NaNO₂ + HCl → HNO₂
Primary Aliphatic Amines:
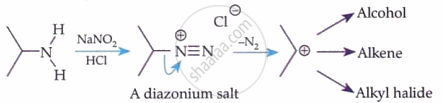
Primary Aromatic Amines (Diazotisation):
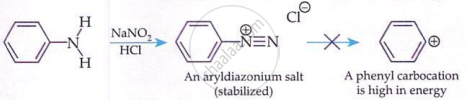
'Aromatic diazonium salts are stable due to delocalisation of the +ve charge in the benzene ring.
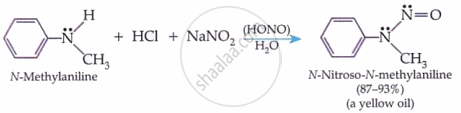
Secondary Amines (both aliphatic and aromatic):
\[ \underset{\text{Dimethylamine}}{(\mathrm{CH}_3)_2 \ddot{\mathrm{N}}\mathrm{H}} + \mathrm{HCl} + \mathrm{NaNO}_2 \xrightarrow{\mathrm{H}_2\mathrm{O}} [(\mathrm{HONO})] \underset{\substack{\text{N-Nitrosodimethylamine} \\ \text{(a yellow oil)}}}{(\mathrm{CH}_3)_2 \ddot{\mathrm{N}} - \ddot{\mathrm{N}} = \mathrm{O}} \]
Tertiary Aliphatic Amines:
\[ \underset{\text{Tertiary aliphatic amine}}{2\mathrm{R}_3\mathrm{N}} + \mathrm{HX} + \mathrm{NaNO}_2 \rightleftharpoons \underset{\text{Amine salt}}{\mathrm{R}_3\overset{+}{\mathrm{N}}\mathrm{H} \text{ } \mathrm{X}^-} + \underset{\text{N-Nitrosoammonium compound}}{\mathrm{R}_3\overset{+}{\mathrm{N}}-\ddot{\mathrm{N}}=\mathrm{O}\mathrm{X}^-} \]
Tertiary Aromatic Amines:
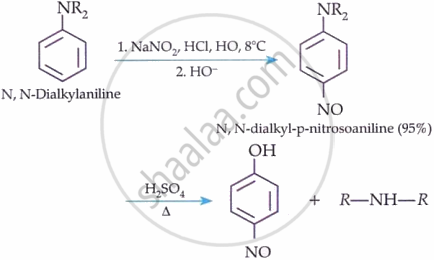
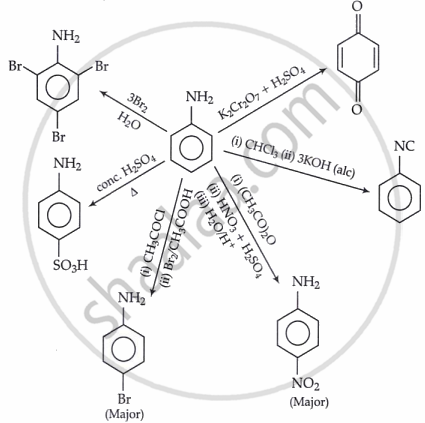
Electrophilic substitution reaction of arylamines:
Reduction of Alkyl Nitriles (Nitrile Reduction):
\[ \mathrm{R} - \mathrm{C} \equiv \mathrm{N} \xrightarrow[\mathrm{LiAlH}_4 \text{ or } \mathrm{Na} / \mathrm{C}_2\mathrm{H}_5\mathrm{OH}]{\text{Raney Ni} / \mathrm{H}_2} \mathrm{R} - \mathrm{CH}_2\mathrm{NH}_2 \]
Reduction of Amides:
\[ \mathrm{R} - \mathrm{CONH}_2 \xrightarrow{\mathrm{LiAlH}_4 / \text{ether}} \mathrm{R} - \mathrm{CH}_2\mathrm{NH}_2 \]
Reduction of Oximes:
\[ \mathrm{R} - \mathrm{CH} = \mathrm{NOH} + 4[\mathrm{H}] \xrightarrow[\mathrm{LiAlH}_4]{\mathrm{Na} / \mathrm{C}_2\mathrm{H}_5\mathrm{OH}} \mathrm{R} - \mathrm{CH}_2\mathrm{NH}_2 + \mathrm{H}_2\mathrm{O} \]
Reductive Amination (from Aldehydes/Ketones):
\[ \mathrm{R} - \mathrm{CH} = \mathrm{O} \xrightarrow{\mathrm{NH}_2\mathrm{OH}} \mathrm{R} - \mathrm{CH} = \mathrm{N} - \mathrm{OH} \xrightarrow{\mathrm{LiAlH}_4} \mathrm{R} - \mathrm{CH}_2 - \mathrm{NH}_2 \]
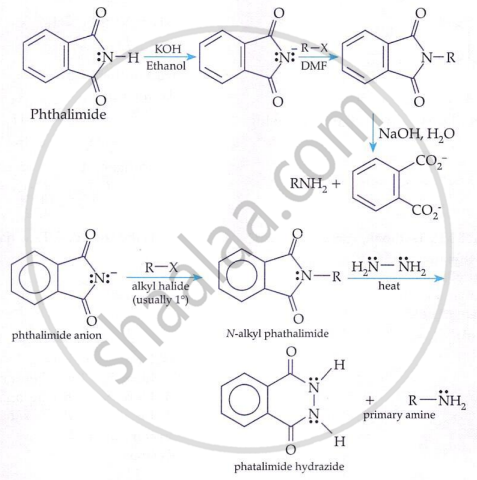
Gabriel Phthalimide Reaction (for 1° amines only):
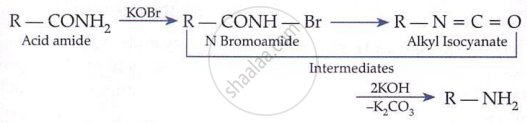
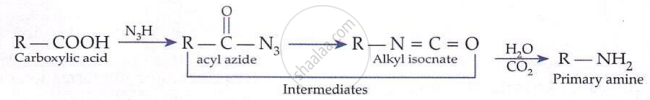
Hoffmann Bromamide Degradation (gives 1° amine with one less carbon):
\[ \underset{\text{Amide}}{\mathrm{RCONH}_2} + \mathrm{Br}_2 + 4\mathrm{KOH} \longrightarrow \underset{1^\circ \text{ amine}}{\mathrm{RNH}_2} + 2\mathrm{KBr} + 2\mathrm{H}_2\mathrm{O} + \mathrm{K}_2\mathrm{CO}_3 \]
Mechanism:
Important Questions [37]
- The Compound ‘B’ is
- Explain Basic Nature of Amines.
- Identify the Weakest Base Amongst the Following
- Among dimethylamine (pKb = 3.27) and diethylamine (pKb = 3.0), which one is more basic?
- How Are 1 - Nitropropane, 2-nitropropane and 2-methyl 2- Nitropropane Are Distinguished from Each Other Using Nitrous Acid?
- Primary and Secondary Nitroalcanes Containing α - H Atom Show Property of
- What is the Action of Nitrous Acid on Primary Nitroalkane?
- When Primary Amine Reacts with Chcl3 in Alcoholic Koh, the Product is
- What is the Action of Benzene Sulphonyl Chloride on Primary, Secondary and Tertiary Amines?
- What is the Action of Nitrous Acid On Secondary Nitroalkane?
- What is the Action of Nitrous Acid on Tertiary Nitroalkane
- What is the action of the following reagents on ethyl amine? Chloroform and caustic potash
- Which of the following amines yields foul smelling product with holoform and alcoholic KOH?
- What will be the action of the mixture of sodium nitrite and dilute hydrochloric acid on aniline
- What will be the action of the mixture of sodium nitrite and dilute hydrochloric acid on triethyl amine
- The amine which reacts with nitrous acid to give yellow oily compound is
- What will be the action of the mixture of sodium nitrite and dilute hydrochloric acid on diethyl amine
- What is the action of the following reagent on ethyl amine? Nitrous acid
- What will be the action of the mixture of sodium nitrite and dilute hydrochloric acid on ethyl amine
- What is the Action of Acetic Anhydride on Ethylamine
- How is Chlorobenzene Prepared from Aniline?
- What is the Action of the Following Reagents on Aniline? Acetic Anhydride
- What is the Action of the Following Reagents on Aniline? Hot and Conc. Sulphuric Acid
- What is the Action of Acetic Anhydride on Diethylamine
- What is the Action of the Following Reagents on Aniline?
- Write a Short Note on Hoffmann Bromamide Degradation.
- Identify the compounds 'A' and 'B' in the following equation
- Write the chemical equation involved in the following reaction: Hoffmann-bromamide degradation reaction
- Write a short note on the following: Hoffmann’s bromamide reaction
- How is Ethyl Amine Prepared from Methyl Iodide?
- Explain the Mechanism of Action of Hydroiodic Acid on 3-methylbutan-2-ol.
- Mention 'Two' Uses of Propan-2-one.
- Write the name of reduction product formed when ethyl cyanide is treated with sodium and alcohol.
- Identify 'A' and 'B' in the Following Reaction and Rewrite the Complete Reaction :
- Write reactions to bring about the following conversions. Acetamide to Ethylamine
- Write the name of the product formed by the action of LiAlH4/ether on acetamide.
- How Are Propan-1-amine and Propan-2-amine Prepared from Oxime?
Concepts [12]
- Concept of Amines
- Classification of Amines
- Structure of Amines
- Physical Properties of Amines
- Uses of Amines
- Identification of Primary, Secondary and Tertiary Amines
- Secondary and Tertiary Amines
- Chemical Reactions of Amines - Carbylamine Reaction
- Chemical Reactions of Amines - Reaction with Nitrous Acid
- Chemical Reactions of Amines - Reaction with Arylsulphonyl Chloride
- Chemical Reactions of Amines - Electrophilic Substitution
- Preparation of Amines
