Definitions [37]
Define isomorphism.
Two or more substances having the same crystal structures are called isomorphous substances, and the phenomenon is called isomorphism. For example, NaF and MgO, NaNO3 and CaCO3 are isomorphous pairs and have the same atomic ratios, 1 : 1 and 1 : 1 : 3, respectively, of the constituent atoms.
Define unit cell.
A basic repeating structural unit of a crystalline solid is called a unit cell.
Define Anisotropy.
The ability of crystalline solids to change values of physical properties when measured in different directions is called anisotropy.
The three-dimensional arrangement of constituent particles of a crystalline solid in space, in which each particle is depicted as a point, is known as a crystal lattice.
A unit cell is the smallest group of lattice points that, when repeated in all directions, will develop the entire lattice.
Packing efficiency is the ratio of volumes occupied by atoms in unit cell to the total volume of the unit cell. It is also known as the packing fraction or the density of packing.
The defects are basically irregularities in the arrangement of constituent particles. These irregularities are called crystal defects.
A pure semiconductor with very low but finite electrical conductivity is called an intrinsic semiconductor.
The process by which impurities are introduced into semiconductors to enhance their conductivity is called doping.
A doped semiconductor, having higher conductivity than a pure intrinsic semiconductor, is an extrinsic semiconductor.
A solid in which the constituent particles are arranged in a regular and periodic three-dimensional pattern is called a crystalline solid.
A solid in which the constituent particles are irregularly arranged and lack long-range order is called an amorphous solid.
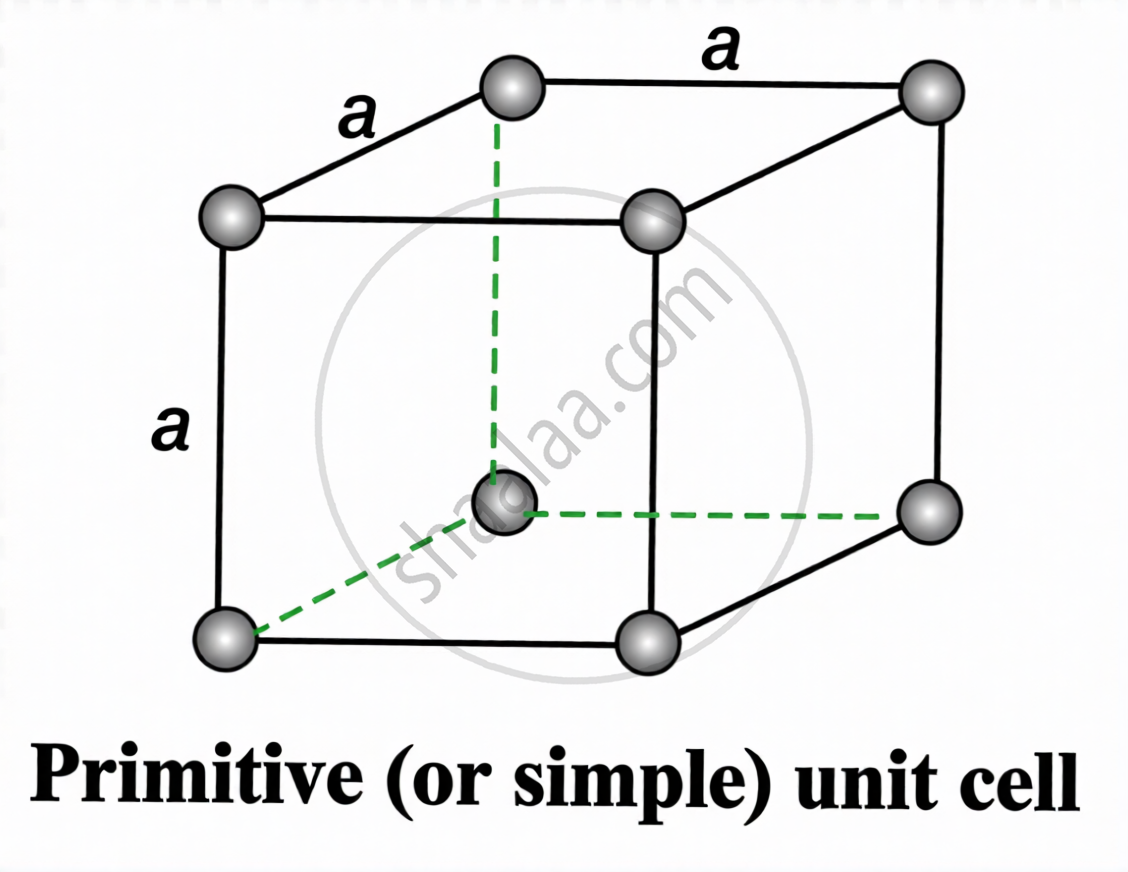
A unit cell in which particles are present only at the eight corners of a cube is called a primitive or simple cubic unit cell (sc).
\[a=2r\quad;\quad r=\frac{a}{2}\]
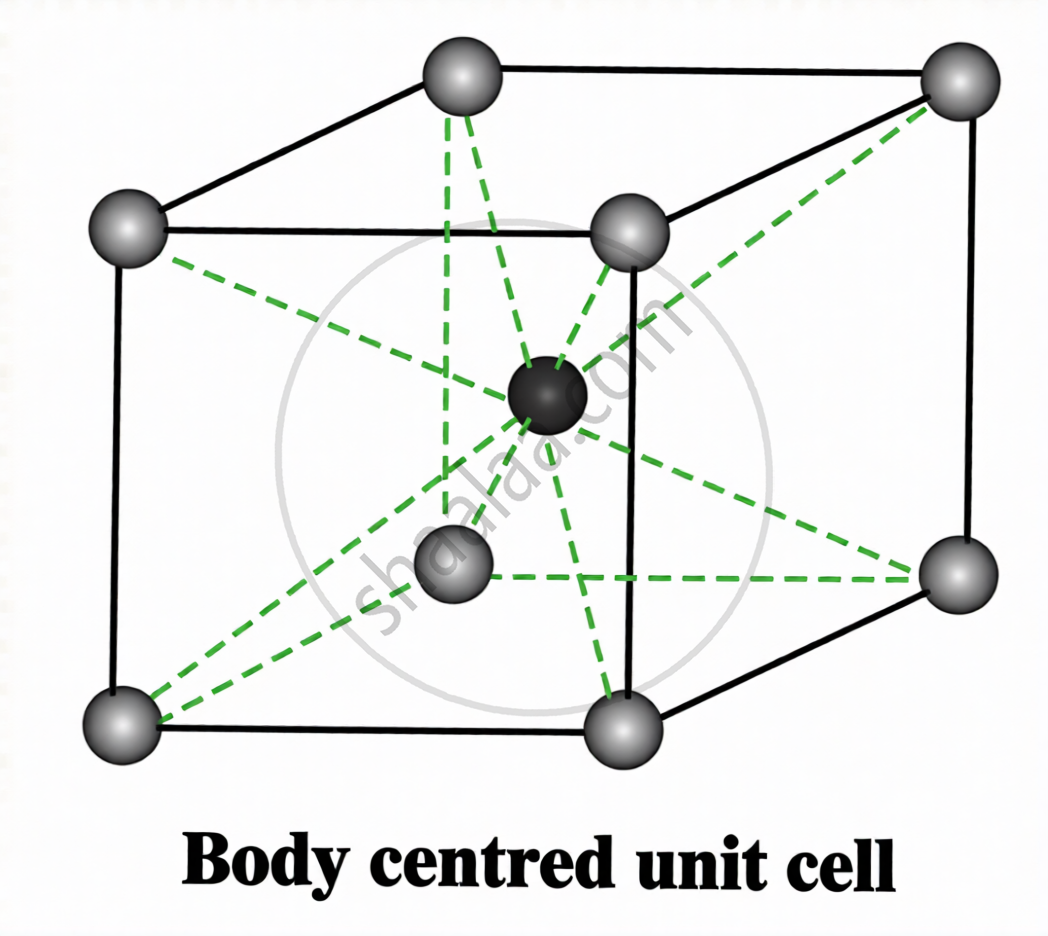
A unit cell in which particles are present at the eight corners and one particle at the centre of the cube is called a body-centred cubic unit cell (bcc).
\[\sqrt3a=4r\quad;\quad r=\frac{\sqrt3a}{4}\]
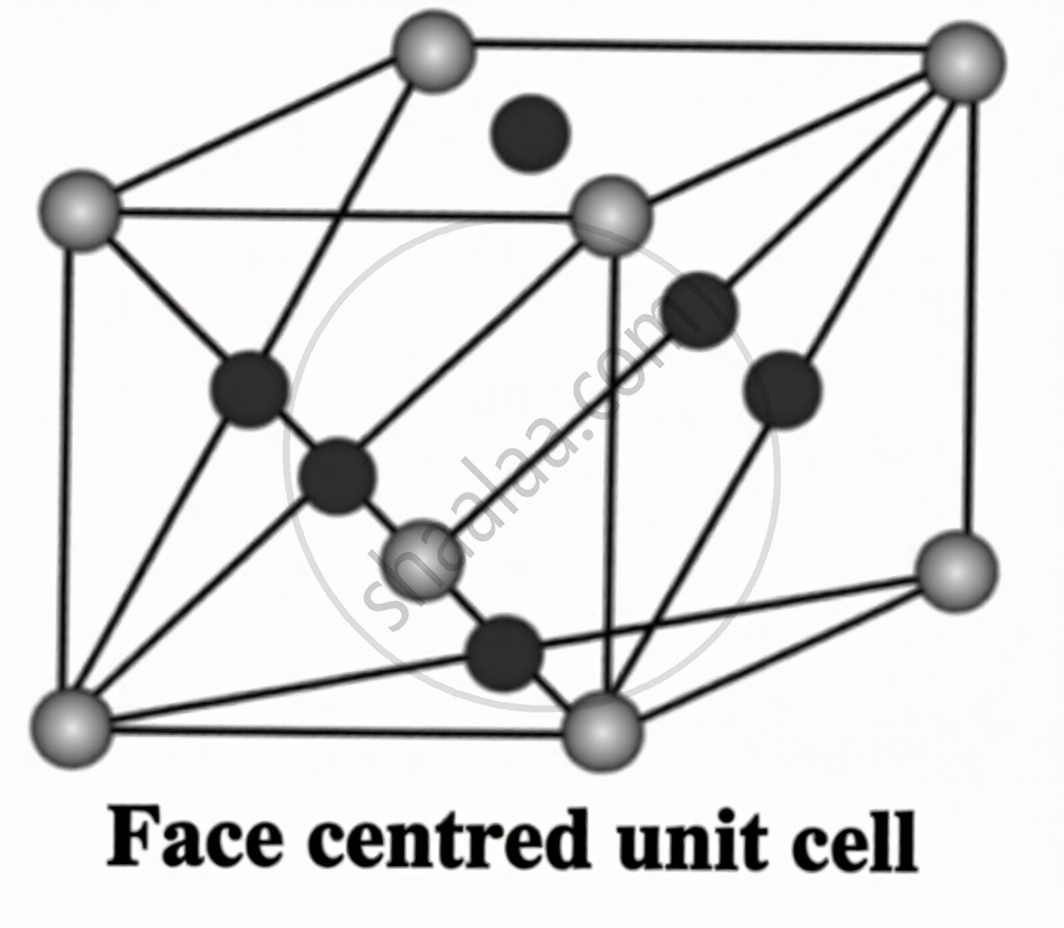
A unit cell in which particles are present at the eight corners and at the centre of each of the six faces of the cube is called a face-centred cubic unit cell (fcc).
\[\sqrt2a=4r\quad;\quad r=\frac{a}{2\sqrt2}\]
The number of nearest neighbouring particles surrounding a given particle in a crystal is called the coordination number.
The empty space present in a crystal lattice not occupied by particles is called void space.
The defect in ionic solids where equal number of cations and anions are missing from their lattice sites is called Schottky defect.
The defect in ionic solids where an ion leaves its normal lattice site and occupies an interstitial position is called Frenkel defect.
A regular three-dimensional geometrical arrangement of points in space representing the positions of particles in a crystal is called a lattice.
The magnetic property of a substance strongly attracted by magnetic field and retaining magnetism is called ferromagnetism.
An impurity added to a semiconductor to increase its conductivity is called dopant.
The defect in which some lattice points are vacant due to missing atoms or ions is called vacancy defect.
The lower energy band filled with electrons is called valence band.
The highest energy band containing electrons in a solid is called conduction band.
The defect in which impurity atoms replace host atoms at lattice sites is called substitutional impurity defect.
The defect in which impurity atoms occupy interstitial spaces in the lattice is called interstitial impurity defect.
A crystalline solid composed of oppositely charged ions held together by electrostatic forces of attraction is called an ionic solid.
A solid whose constituent particles are molecules held together by intermolecular forces is called a molecular solid.
A crystalline solid formed by metal atoms held together by metallic bonds due to the presence of delocalised electrons is called a metallic solid.
The defect caused by the presence of foreign atoms in a crystal lattice is called impurity defect.
A solid having high electrical conductivity is called a conductor.
A solid having very low electrical conductivity is called an insulator.
A solid having electrical conductivity intermediate between conductor and insulator is called a semiconductor.
The magnetic property of a substance due to presence of unpaired electrons weakly attracted by magnetic field is called paramagnetism.
The process of adding controlled impurity to increase conductivity of a semiconductor is called doping.
The energy difference between valence band and conduction band is called band gap.
Formulae [7]
Packing fraction or Packing efficiency = \[\frac{\text{Total volume of spheres}}{\text{Volume of the unit cell}}\times100\]
Volume = a3
\[\rho=\frac{m\times n}{a^3}\]
\[m=\frac{M}{N_A}\]
\[\rho=\frac{nM}{a^3N_A}\]
\[V=\frac{4}{3}\pi r^3\]
\[\text{Packing efficiency}=\frac{\text{Volume occupied by particles in unit cell}}{\text{Total volume of unit cell}}\times100\]
Theorems and Laws [1]
Name the law or principle to which the following observations confirm:
When 9650 coulombs of electricity is passed through a solution of copper sulphate, 3.175 g of copper is deposited on the cathode (at. wt. of Cu = 63.5).
Faraday’s first law of electrolysis: The mass of a substance deposited or liberated at an electrode is directly proportional to the quantity of electricity passed through the electrolyte.
Given: Charge passed = 9650 C
Atomic mass of Cu = 63.5
Valency of Cu in CuSO4 = 2
Equivalent mass of Cu = `63.5/2` = 31.75
Now, Mass deposited = `9650/96500 xx 31.75`
= 0.1 × 31.75
= 3.175 g
Key Points
| Property | Crystalline Solids | Amorphous Solids |
|---|---|---|
| Arrangement of particles | Regular, ordered | Random, disordered |
| Melting point | Sharp | Not sharp (melts over a range) |
| Optical behaviour | Anisotropy (properties differ with direction) | Isotropy (same in all directions) |
| Examples | NaCl, Fe | Glass, Rubber |
Amorphous solids are also known as supercooled liquids.
Isomorphism and Polymorphism:
| Term | Meaning | Example |
|---|---|---|
| Isomorphism | Two or more substances having the same crystal structure (same atomic ratio) | NaF & MgO (1:1); NaNO₃ & CaCO₃ (1:1:3) |
| Polymorphism | A single substance existing in two or more crystalline forms | Calcite & aragonite (CaCO₃); α-quartz, β-quartz, cristobalite (SiO₂) |
Polymorphism occurring in elements is called allotropy.
| Property | Ionic Solids | Covalent Network Solids | Molecular Solids | Metallic Solids |
|---|---|---|---|---|
| Particles | Cations and anions | Covalently bonded atoms | Mono/polyatomic molecules | Metal ions in a sea of electrons |
| Forces | Electrostatic | Covalent bonds | London, dipole-dipole, H-bonding | Metallic bonds |
| Hardness | Hard and brittle | Very hard | Soft | Soft to very hard |
| Melting point | 600–3000°C | 1200–4000°C | Low (−272 to 400°C) | −39 to 3400°C |
| Conductivity | Poor (solid); good (molten/aqueous) | Poor (except graphite; diamond conducts heat) | Poor | Good conductor of heat and electricity |
| Examples | NaCl, CaF₂ | Diamond, silica | Ice, benzoic acid | Na, Mg, Cu, Au |
Types of Unit Cells:
| Type | Location of Particles | Atoms per Unit Cell |
|---|---|---|
|
Simple / Primitive
|
Corners only | (1/8 × 8) = 1 |
|
Body-centred (bcc)
|
Corners + 1 at body centre | (1/8 × 8) + 1 = 2 |
|
Face-centred (fcc)
|
Corners + 1 at each face centre | (1/8 × 8) + (1/2 × 6) = 4 |
| Base-centred | Corners + centres of two opposite faces | (1/8 × 8) + (1/2 × 2) = 2 |
Seven Crystal Systems:
| System | Intercepts | Angles | Bravais Lattices |
|---|---|---|---|
| Cubic | a = b = c | α = β = γ = 90° | Primitive, FCC, BCC (3) |
| Tetragonal | a = b ≠ c | α = β = γ = 90° | Primitive, BCC (2) |
| Orthorhombic | a ≠ b ≠ c | α = β = γ = 90° | Primitive, FCC, BCC, End-centred (4) |
| Monoclinic | a ≠ b ≠ c | α = γ = 90°, β ≠ 90° | Primitive, End-centred (2) |
| Triclinic | a ≠ b ≠ c | α ≠ β ≠ γ ≠ 90° | Primitive (1) |
| Hexagonal | a = b ≠ c | α = β = 90°, γ = 120° | Primitive (1) |
| Rhombohedral | a = b = c | α = β = γ ≠ 90° | Primitive (1) |
Close Packing of Spheres:
| Packing | Type | Coordination Number |
|---|---|---|
| 1D (linear) | Spheres in a row | 2 |
| 2D square (AAAA) | Square close packed | 4 |
| 2D hexagonal (ABAB) | Hexagonal close packed | 6 |
| 3D simple cubic (AAAA) | e.g., Polonium | 6 |
| 3D hcp (ABAB) | e.g., Mg, Zn | 12 |
| 3D ccp/fcc (ABCABC) | e.g., Cu, Ag | 12 |
Both hcp and ccp have the same coordination number (12) and same packing efficiency (74%).
Types of Voids:
| Void | Surrounded by | Size (relative to r) |
|---|---|---|
| Trigonal / Triangular | 3 spheres | 0.15 r |
| Tetrahedral | 4 spheres | 0.225 r |
| Octahedral | 6 spheres | 0.414 r |
Increasing order of void size: trigonal < tetrahedral < octahedral
Number of Voids (in hcp/ccp):
- Tetrahedral voids = 2N
- Octahedral voids = N (N = number of close-packed spheres)
- Octahedral voids = half of tetrahedral voids
Solids are classified into three groups based on conductivity:
| Type | Conductivity (Ohm⁻¹ m⁻¹) | Band Gap | Reason | Examples |
|---|---|---|---|---|
| Metallic conductors | 10⁴ – 10⁷ (very high) | No band gap (overlapping s & p bands) | Motion of electrons | Cu, Al, Ag |
| Insulators | 10⁻²⁰ – 10⁻¹⁰ (very low) | Large (forbidden zone) | Electrons cannot cross | Diamond, wood, rubber |
| Semiconductors | 10⁻⁶ – 10⁴ (moderate) | Small | Motion of interstitial electrons/holes | Si, Ge |
Conductivity of metals decreases with increase in temperature; conductivity of semiconductors increases with temperature.
| Type of Crystalline Solid | Constituent Particles | Type of Bonding / Forces | Main Properties | Examples |
|---|---|---|---|---|
| Ionic Solids | Cations and anions | Electrostatic force of attraction (ionic bond) | Hard and brittle; high melting points; non-conductors in solid state; conduct electricity when molten or dissolved in water | NaCl, KCl, K₂SO₄, CaF₂ |
| Covalent Network Solids | Atoms | Continuous covalent bonds (3D network) | Very hard; very high melting and boiling points; poor conductors of electricity (except graphite) | Diamond, SiO₂ (quartz), BN, SiC |
| Molecular Solids | Molecules | Weak intermolecular forces (dipole-dipole, London forces, hydrogen bonding) | Soft; low melting points; poor conductors of electricity | Ice (H₂O), CO₂, CH₄, NH₃ |
| Metallic Solids | Metal atoms (positive ions in sea of electrons) | Metallic bond (delocalised electrons) | Malleable, ductile; good conductors of heat and electricity | Na, Fe, Cu, Ag, Au |
| S. No. | Crystal System | Axial Lengths (a, b, c) | Axial Angles (α, β, γ) | Types of Bravais Lattices (Unit Cells) | Total Lattices |
|---|---|---|---|---|---|
| 1 | Cubic | a = b = c | α = β = γ = 90° | (i) Simple (Primitive) (ii) Body-Centred (BCC) (iii) Face-Centred (FCC) | 3 |
| 2 | Tetragonal | a = b ≠ c | α = β = γ = 90° | (i) Simple (Primitive) (ii) Body-Centred | 2 |
| 3 | Orthorhombic | a ≠ b ≠ c | α = β = γ = 90° | (i) Simple (Primitive) (ii) Body-Centred (iii) Face-Centred (iv) Base-Centred | 4 |
| 4 | Monoclinic | a ≠ b ≠ c | α = γ = 90°, β ≠ 90° | (i) Simple (Primitive) (ii) Base-Centred | 2 |
| 5 | Triclinic | a ≠ b ≠ c | α ≠ β ≠ γ ≠ 90° | (i) Simple (Primitive) | 1 |
| 6 | Rhombohedral (Trigonal) | a = b = c | α = β = γ ≠ 90° | (i) Simple (Primitive) | 1 |
| 7 | Hexagonal | a = b ≠ c | α = β = 90°, γ = 120° | (i) Simple (Primitive) | 1 |
| Type of Solid | Electron Arrangement | Behaviour in Magnetic Field | Magnetic Nature | Examples |
|---|---|---|---|---|
| Diamagnetic Solids | All electrons are paired | Weakly repelled by magnetic field | No permanent magnetic moment; magnetism cancelled due to pairing of electrons | N₂, F₂, NaCl, H₂O, Benzene |
| Paramagnetic Solids | One or more unpaired electrons | Weakly attracted by magnetic field | Temporary magnetism; show magnetism only in presence of external magnetic field | O₂, Cu²⁺, Fe³⁺, Cr³⁺ |
| Ferromagnetic Solids | Large number of unpaired electrons | Strongly attracted by magnetic field | Can be permanently magnetised; retain magnetism even after removal of field | Fe, Co, Ni, Gd, CrO₂ |
| Main Type | Sub-Type | Description | Effect on Density | Electrical Neutrality | Examples / Notes |
|---|---|---|---|---|---|
| Stoichiometric Defects | Vacancy Defect | Some lattice sites are vacant due to missing particles | Decreases (mass decreases, volume same) | Maintained | Common in ionic solids |
| Self-Interstitial Defect | Extra particle occupies interstitial space | Increases (mass increases, volume same) | Maintained | Seen in elemental solids | |
| Schottky Defect | Equal number of cations and anions are missing from lattice sites | Decreases | Maintained | NaCl, KCl, AgBr | |
| Frenkel Defect | Ion leaves normal lattice site and occupies interstitial site | No change | Maintained | ZnS, AgCl, AgBr | |
| Impurity Defects | Substitutional Impurity | Foreign atom replaces host atom in lattice | May change | Maintained | Brass (Cu–Zn alloy) |
| Interstitial Impurity | Foreign atom occupies interstitial space | May change | Maintained | Stainless steel (Fe–C) | |
| Vacancy due to Aliovalent Impurity | Different valency impurity creates vacancies to maintain neutrality | Decreases | Maintained | Sr²⁺ added to NaCl | |
| Non-Stoichiometric Defects | Metal Deficiency Defect | Missing metal ions; charge balanced by higher oxidation state | Slight change | Maintained | NiO → Ni₀.₉₇O |
| Metal Excess Defect (Interstitial) | Extra metal ions occupy interstitial sites | Increases | Maintained | ZnO | |
| Metal Excess Defect (F-centre) | Anion vacancy occupied by electron | No major change | Maintained | NaCl (yellow colour) |
Important Questions [25]
- Distinguish between crystalline solid and amorphous solid.
- Define isomorphism.
- An Ionic Compound Crystallises in FCC Type Structure with ‘A’ Ions at the Centre of Each Face and ‘B’ Ions Occupying Corners of the Cube. the Formula of Compound is
- Classify the Following Solids into Different Types
- Distinguish Between Crystalline Solids and Amorsphous Solids.
- Classify the Following Molecular Solids into Different Types: Hcl Co2 Solid Ice So2
- Define Anisotropy.
- The Major Binding Force in Diamond is
- Define unit cell.
- Explain ionic solids are hard and brittle.
- An element with molar mass 27 g/mol forms a cubic unit cell with edge length of 405 p.m. If the density of the element is 2.7 g/cm3, what is the nature of the cubic unit cell?
- Obtain the relationship between the density of a substance and the edge length of the unit cell.
- The co-ordination number of atoms in body centred cubic structure (bcc) is ______.
- Gold crystallises into face-centred cubic cells. The edge length of a unit cell is 4.08 × 10–8 cm. Calculate the density of gold. [Molar mass of gold = 197 g mol–1]
- Silver crystallizes in the fcc structure. If the edge length of the unit cell is 400 pm, calculate the density of silver (Atomic mass of Ag = 108).
- The number of particles present in Face Centred Cubic Unit cell is/are ______.
- The coordination number of atoms in bcc crystal lattice is ______.
- Predict the type of cubic lattice of a solid element having edge length of 400 pm and density is 6.25 g/ml.(Atomic mass of element = 60)
- The density of iron crystal is 8.54-gram cm–3. If the edge length of the unit cell is 2.8 A° and atomic mass is 56 gram mol–1
- Cu crystallizes in fcc unit cell with edge length of 495 pm. What is the radius of Cu atom?
- The relation between the radius of the sphere and the edge length in the body-centred cubic lattice is given by the formula ______.
- Write the consequences of Schottky defect with reasons.
- Explain metal deficiency defect with example.
- Explain the following term: Substitutional impurity defect
- Explain the following term: Interstitial impurity defect