Advertisements
Advertisements
Question
What is the action of Benzene Sulphonyl Chloride on primary, secondary and tertiary
amines?
Advertisements
Solution
Reaction of primary amine with benzene sulphonyl chloride gives corresponding
N-alkyl benzene sulphonylamide.
Owing to the presence of strong electron withdrawing sulphonyl group, hydrogen
attached to nitrogen atom in N-alkyl benzene sulphonylamide is strongly acidic
and dissolves in aqueous KOH. On acidification of this solution, insoluble amide
gets regenerated.

Reaction of secondary amine with benzene sulphonyl chloride gives N,N-dialkyl
benzene sulphonylamide.
N,N-dialkyl benzene sulphonylamide does not contain acidic hydrogen. Hence, it is not acidic and is insoluble in aqueous KOH. It is also insoluble in acid.

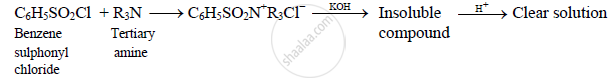
Due to absence of hydrogen atom directly attached to the nitrogen atom, tertiary
amines do not react with benzene sulphonyl chloride. The unreacted tertiary
amine is insoluble in aqueous KOH but soluble in acid.
APPEARS IN
RELATED QUESTIONS
What is the action of nitrous acid on primary nitroalkane?
Distinguish between the following pairs of compounds: Aniline and N-methylaniline
Describe a method for the identification of primary, secondary and tertiary amines. Also write chemical equations of the reactions involved.
When primary amine reacts with CHCl3 in alcoholic KOH, the product is _______.
(A) aldehyde
(B) alcohol
(C) cyanide
(D) an isocyanide
What is the action of nitrous acid on secondary nitroalkane?
What is the action of nitrous acid on tertiary nitroalkane
Describe Hinsberg method for the identification of primary, secondary and tertiary amines. Also write the chemical equations of the reactions involved.
Identify the compound that will react with Hinsberg’s reagent to give a solid which dissolves in alkali.
Identify the compound that will react with Hinsberg’s reagent to give a solid which dissolves in alkali.
Arrange the following in the increasing order of their boiling points:
A: Butanamine
B: N, N-Dimethylethanamine
C: N- Etthylethanaminamine
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Tertiary amines are more basic than corresponding secondary and primary amines in gaseous state.
Reason (R): Tertiary amines have three alkyl groups which cause +I effect.
Select the most appropriate answer from the options given below:
Compound A is converted to B on reaction with CHCl3 and KOH. The compound B is toxic and can be decomposed by C. A, B and C respectively are ______.
Isocyanide reaction involves the intermediate formation of ______.
The presence of primary amines can be confirmed by ______.
Which of the following is not a correct statement for primary aliphatic amines?
Match List I with List II.
| List I | List II | ||
| (A) | Benzenesulphonyl chloride | (I) | Test for primary amines |
| (B) | Hoffmann bromamide reaction | (II) | Anti Saytzeff |
| (C) | Carbylamine reaction | (III) | Hinsberg reagent |
| (D) | Hoffmann orientation | (IV) | Known reaction of Isocyanates |
Choose the correct answer from the options given below:
A compound 'X' on treatment with Br2/NaOH, provided C3H9N, which gives positive carbylamine test. Compound 'X' is ______.
In the reaction \[\ce{C6H5NH2 + CHCl3 + 3KOH -> A + 3B + 3C}\] the product A is:
Carbylamine test is the distinguishing test for ______.
Among dimethylamine (pKb = 3.27) and diethylamine (pKb = 3.0), which one is more basic?
Which of the following reactions will not give a primary amine as the product?
