Advertisements
Online Mock Tests
Chapters
2: Chemical Bonding
3: Acids, Bases and Salts
4: Analytical Chemistry
5: Mole concept and Stoichiometry
6: Electrolysis
7: Metallurgy
8: Study of Compounds A. Hydrogen Chloride
9: Study of Compounds B. Ammonia
10: Study of Compounds C. Nitric Acid
▶ 11: Study of Compounds D. Sulphuric Acid
12: Organic Chemistry
13: Practical Work
![S.P. Singh solutions for कन्साइस केमिस्ट्री [अंग्रेजी] कक्षा १० आईसीएसई chapter 11 - Study of Compounds D. Sulphuric Acid S.P. Singh solutions for कन्साइस केमिस्ट्री [अंग्रेजी] कक्षा १० आईसीएसई chapter 11 - Study of Compounds D. Sulphuric Acid - Shaalaa.com](/images/concise-chemistry-english-class-10-icse_6:7b16b8cbeb0b4ebab074df2efa4502e0.jpg)
Advertisements
Solutions for Chapter 11: Study of Compounds D. Sulphuric Acid
Below listed, you can find solutions for Chapter 11 of CISCE S.P. Singh for कन्साइस केमिस्ट्री [अंग्रेजी] कक्षा १० आईसीएसई.
S.P. Singh solutions for कन्साइस केमिस्ट्री [अंग्रेजी] कक्षा १० आईसीएसई 11 Study of Compounds D. Sulphuric Acid Intext Questions [Page 175]
Comment, sulphuric acid is referred to as King of chemicals.
Comment, sulphuric acid is referred to as oil of vitriol.
Sulphuric acid is manufactured by the contact process.
Give two balanced equations to obtain SO2 in this process.
Sulphuric acid is manufactured by the contact process.
Give the conditions for the oxidation of SO2.
Sulphuric acid is manufactured by the contact process.
Name the catalyst used.
Sulphuric acid is manufactured by the contact process.
Why H2SO4 is not obtained by directly reacting SO3 with water?
Sulphuric acid is manufactured by the contact process.
Name the chemical used to dissolve SO3 and also name the product formed. Give all the main reactions to this process.
Why the impurity of arsenic oxide must be removed before passing the mixture of SO2 and air through the catalytic chamber?
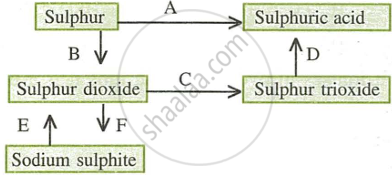
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
S.P. Singh solutions for कन्साइस केमिस्ट्री [अंग्रेजी] कक्षा १० आईसीएसई 11 Study of Compounds D. Sulphuric Acid EXERCISE-11 [Pages 179 - 181]
Why is water not added to concentrated H2SO4 in order to dilute it?
Give two balanced reactions of the type to show the following property of sulphuric acid:
Acidic nature
Give two balanced reactions of the type to show the following property of sulphuric acid:
Oxidising agent
Give two balanced reactions of the type to show the following property of sulphuric acid:
Dehydrating nature
Give two balanced reactions of the type to show the following property of sulphuric acid:
Non-volatile nature
Give a chemical test to distinguish between dilute sulphuric acid and dilute hydrochloric acid (using lead nitrate solution).
Give a chemical test to distinguish between dilute sulphuric acid and conc. sulphuric acid.
Write a balanced chemical equation:
When hot and concentrated sulphuric acid reacts with the sulphur.
Write a balanced chemical equation:
When hot and concentrated sulphuric acid reacts with the NaOH.
Write a balanced chemical equation:
When hot and concentrated sulphuric acid reacts with the sugar.
Write a balanced chemical equation:
When hot and concentrated sulphuric acid reacts with the carbon.
Write a balanced chemical equation:
When hot and concentrated sulphuric acid reacts with the copper.
Why is concentrated sulphuric acid kept in air tight bottles?
Why is H2SO4 is not used as a drying agent for H2S?
Why is sulphuric acid used in the preparation of HCl and HNO3? Give equations in both cases.
What property of conc. H2SO4 is made use of in the following case? Give an equation for the reaction in this case:
In the production of HCl gas when it reacts with chloride.
What property of conc. H2SO4 is made use of in the following case? Give an equation for the reaction in this case:
In the preparation of CO and HCOOH.
What property of conc. H2SO4 is made use of in the following case? Give an equation for the reaction in this case:
As a source of hydrogen by diluting it and adding a strip of magnesium.
What property of conc. H2SO4 is made use of in the following case? Give an equation for the reaction in this case:
In the preparation of sulphur dioxide by warming a mixture of conc. sulphuric acid and copper turnings.
What property of conc. H2SO4 is made use of in the following case? Give an equation for the reaction in this case:
Hydrogen chloride gas is passed through concentrated sulphuric acid.
What property of conc. H2SO4 is made use of in the following case? Give an equation for the reaction in this case:
It’s reaction with ethanol.
What property of conc. H2SO4 is made use of in the following case? Give an equation for the reaction in this case:
It’s reaction with carbon.
What is the name given to the salts of sulphurous acid?
What is the name given to the salts of sulphuric acid?
Give reasons for the following:
Sulphuric acid forms two types of salts with NaOH.
Give reasons for the following:
A piece of wood becomes black when concentrated sulphuric acid is poured on it.
Give reasons for the following:
Brisk effervescence is seen when oil of vitriol is added to sodium carbonate.
Copy and complete the following table:
| Column 1 | Column 2 | Column 3 |
| Substance reacted with acid | Dilute or concentrated acid | Gas |
| Hydrogen | ||
| Carbon dioxide | ||
| Only chlorine |
The following is the typical property of dilute acid. Complete it by inserting suitable words:
\[\ce{Active metal + Acid -> \underline{\phantom{.......}} + \underline{\phantom{.......}}}\]
The following is the typical property of dilute acid. Complete it by inserting suitable words:
\[\ce{Base + Acid -> \underline{\phantom{.......}} + \underline{\phantom{.......}}}\]
The following is the typical property of dilute acid. Complete it by inserting suitable words:
\[\ce{Carbonate/hydrogen carbonate + Acid -> \underline{\phantom{.......}} + \underline{\phantom{.......}} + \underline{\phantom{.......}}}\]
The following is the typical property of dilute acid. Complete it by inserting suitable words:
\[\ce{Sulphite/hydrogen sulphite + Acid -> \underline{\phantom{.......}} + \underline{\phantom{.......}} + \underline{\phantom{.......}}}\]
The following is the typical property of dilute acid. Complete it by inserting suitable words:
\[\ce{Sulphide + Acid -> \underline{\phantom{.......}} + \underline{\phantom{.......}}}\]
Which property of sulphuric acid accounts for its use as a dehydrating agent?
Concentrated sulphuric acid is both an oxidizing agent and a non-volatile acid. Write one equation. Each illustrates the above-mentioned properties of sulphuric acid.
Some properties of sulphuric acid are listed below. Choose the property A, B, C or D which is responsible for the reactions (i) to (v). Some properties may be repeated:
- Typical acid
- Dehydrating agent
- Non-volatile acid
- Oxidizing agent
- \[\ce{C12H22O11 + nH2SO4 -> 12C + 11H2O + nH2SO4}\]
- \[\ce{S + 2H2SO4 -> 3SO2 + 2H2O}\]
- \[\ce{NaCl + H2SO4 -> NaHSO4 + HCl}\]
- \[\ce{CuO + H2SO4 -> CuSO4 + H2O}\]
- \[\ce{Na2CO3 + H2SO4 -> Na2SO4 + H2O + CO2}\]
Name the acid formed when sulphur dioxide dissolves in water.
Name the gas released when sodium carbonate is added to a solution of sulphur dioxide.
2008
Dilute sulphuric acid will produce a white precipitate when added to a solution of ______.
Copper nitrate
Zinc nitrate
Lead nitrate
Sodium nitrate
Identify the following substance:
Liquid E can be dehydrated to produce ethene.
Copy and complete the following table relating to an important industrial process and its final output.
| Name of process | Inputs | Catalyst | Equation for catalyzed reaction output |
| Contact process | Sulphur dioxide + oxygen |
Making use only of substances chosen from those given below:
(Dilute sulphuric acid, sodium carbonate, Zinc, sodium sulphite, Lead, calcium carbonate)
Give the equation for the reaction by which you could obtain hydrogen.
Making use only of substances chosen from those given below:
(Dilute sulphuric acid, sodium carbonate, Zinc, sodium sulphite, Lead, calcium carbonate)
Give the equation for the reaction by which you could obtain sulphur dioxide.
Making use only of substances chosen from those given below:
(Dilute sulphuric acid, sodium carbonate, Zinc, sodium sulphite, Lead, calcium carbonate)
Give the equation for the reaction by which you could obtain carbon dioxide.
Making use only of substances chosen from those given below:
(Dilute sulphuric acid, sodium carbonate, Zinc, sodium sulphite, Lead, calcium carbonate)
Give the equation for the reaction by which you could obtain zinc carbonate (two steps required).
What property of conc. H2SO4 is used in the action when sugar turns black in its presence?
What is the property of concentrated sulphuric acid (H2SO4) which allows it to be used in the preparation of hydrogen chloride (HCl) and nitric acid (HNO3)?
Write the equation for dil. H2SO4 and barium chloride.
Write the equation for dil. H2SO4 and sodium sulphide.
2009
Name the gas evolved [formula is not acceptable]. The gas that can be oxidised to sulphur.
2010
Write the equation of the following reaction:
Sulphur is heated with concentrated sulphuric acid.
Write the reaction of conc. H2SO4 with sugar.
Give a balanced equation for the conversion of zinc oxide to zinc sulphate.
Select the correct answer from the options:
The solution which liberates sulphur dioxide gas, from sodium sulphite.
Sodium hydroxide solution
A weak acid
Dilute sulphuric acid
2011
What would you observe in the following case:
Sugar crystals are added to a hard glass test tube containing concentrated sulphuric acid.
Choose the correct answer from the options:
The gas evolved when dilute sulphuric acid reacts with iron sulphide.
Sulphur dioxide
Carbon dioxide
Hydrogen sulphide
Nitrogen dioxide
Sulphur trioxide
Vapour of sulphuric acid
Write balanced chemical equations for the action of dilute sulphuric acid on sodium sulphite.
Give a balanced equation for the manufacture of sulphuric acid by the contact process.
State the property of sulphuric acid shown by the reaction of conc. sulphuric acid when heated with potassium nitrate.
Which property of sulphuric acid is shown by the reaction of the concentrated sulphuric acid with carbon?
State the property of sulphuric acid shown by the reaction of conc. sulphuric acid when heated with ethanol.
2012
Name the gas produced on the reaction of dilute sulphuric acid with a metallic sulphide.
Some properties of sulphuric acid are listed below. Choose the role played by sulphuric acid as A, B, C or D which is responsible for the reactions (i) to (v). Some role/s may be repeated.
- Dilute acid
- Dehydrating agent
- Non-volatile acid
- Oxidising agent
- \[\ce{CuSO4 * 5H2O ->[conc. H2SO4] CuSO4 + 5H2O}\]
- \[\ce{S + 2H2SO4 [conc.] -> 3SO2 + 2H2O}\]
- \[\ce{NaNO3 + \underset{[conc.]}{H2SO4} ->[<200^\circ C] NaHSO4 + HNO3}\]
- \[\ce{MgO + H2SO4 -> MgSO4 + H2O}\]
- \[\ce{Zn + 2H2SO4 [conc.] -> ZnSO4 + SO2 + 2H2O}\]
Give a balanced equation for the reaction:
Zinc sulphide and dilute sulphuric acid.
State one appropriate observation when conc. H2SO4 is added to a crystal of hydrated copper sulphate.
In the given equation \[\ce{S + 2H2SO4 -> 3SO2 + 2H2O}\]. Identify the role played by conc. H2SO4.
Non-volatile acid
Oxidising agent
Dehydrating agent
None of the above
State one appropriate observation of the following:
Dehydration of concentrated sulphuric acid with sugar crystals.
Identify the substance underlined:
A dilute mineral acid which forms a white precipitate when treated with barium chloride solution.
2014
Write a balanced equation for the following:
Action of concentrated sulphuric acid on carbon.
Distinguish between the following pair of compounds using the test given with brackets:
Dilute sulphuric acid and dilute hydrochloric acid (using barium chloride solution)
State the conditions required for the following reactions to take place:
The conversion of sulphur dioxide to sulphur trioxide.
Give one equation to show the following property of sulphuric acid:
Dehydrating property
Give one equation to show the following property of sulphuric acid:
Acidic nature
Give one equation to show the following property of sulphuric acid:
As a non-volatile acid
2015
In the contact process for the manufacture of sulphuric acid give the equations for the conversion of sulphur trioxide to sulphuric acid.
Give a balanced chemical equation for the action of sulphuric acid on the following:
Potassium hydrogen carbonate
Give a balanced chemical equation for the action of sulphuric acid on the following:
Sulphur
Identify the acid in the following case:
The acid which produces sugar charcoal from sugar.
Identify the acid in the following case:
The acid on mixing with lead nitrate solution produces a white precipitate, which is insoluble even on heating.
Solutions for 11: Study of Compounds D. Sulphuric Acid
![S.P. Singh solutions for कन्साइस केमिस्ट्री [अंग्रेजी] कक्षा १० आईसीएसई chapter 11 - Study of Compounds D. Sulphuric Acid S.P. Singh solutions for कन्साइस केमिस्ट्री [अंग्रेजी] कक्षा १० आईसीएसई chapter 11 - Study of Compounds D. Sulphuric Acid - Shaalaa.com](/images/concise-chemistry-english-class-10-icse_6:7b16b8cbeb0b4ebab074df2efa4502e0.jpg)
S.P. Singh solutions for कन्साइस केमिस्ट्री [अंग्रेजी] कक्षा १० आईसीएसई chapter 11 - Study of Compounds D. Sulphuric Acid
Shaalaa.com has the CISCE Mathematics कन्साइस केमिस्ट्री [अंग्रेजी] कक्षा १० आईसीएसई CISCE solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. S.P. Singh solutions for Mathematics कन्साइस केमिस्ट्री [अंग्रेजी] कक्षा १० आईसीएसई CISCE 11 (Study of Compounds D. Sulphuric Acid) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. S.P. Singh textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in कन्साइस केमिस्ट्री [अंग्रेजी] कक्षा १० आईसीएसई chapter 11 Study of Compounds D. Sulphuric Acid are Sulphuric Acid, Manufacture of Sulphuric Acid, Physical Properties of Sulphuric Acid, Chemical Properties of Sulphuric Acid, Uses of Sulphuric Acid, Tests for Sulphuric Acid and Sulphates, Sulphuric Acid, Manufacture of Sulphuric Acid, Physical Properties of Sulphuric Acid, Chemical Properties of Sulphuric Acid, Uses of Sulphuric Acid, Tests for Sulphuric Acid and Sulphates, Sulphuric Acid, Manufacture of Sulphuric Acid, Physical Properties of Sulphuric Acid, Chemical Properties of Sulphuric Acid, Uses of Sulphuric Acid, Tests for Sulphuric Acid and Sulphates.
Using S.P. Singh कन्साइस केमिस्ट्री [अंग्रेजी] कक्षा १० आईसीएसई solutions Study of Compounds D. Sulphuric Acid exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in S.P. Singh Solutions are essential questions that can be asked in the final exam. Maximum CISCE कन्साइस केमिस्ट्री [अंग्रेजी] कक्षा १० आईसीएसई students prefer S.P. Singh Textbook Solutions to score more in exams.
Get the free view of Chapter 11, Study of Compounds D. Sulphuric Acid कन्साइस केमिस्ट्री [अंग्रेजी] कक्षा १० आईसीएसई additional questions for Mathematics कन्साइस केमिस्ट्री [अंग्रेजी] कक्षा १० आईसीएसई CISCE, and you can use Shaalaa.com to keep it handy for your exam preparation.
