Advertisements
Advertisements
प्रश्न
Give one equation to show the following property of sulphuric acid:
Dehydrating property
Advertisements
उत्तर
H2SO4 has a great affinity for water, and therefore, it acts as a dehydrating agent.
\[\ce{HCOOH ->[conc.H2SO4] CO + H2O}\]
\[\ce{C2H5OH ->[conc.H2SO4] C2H4 + H2O}\]
संबंधित प्रश्न
Distinguish between the given pairs of compounds using the test given within brackets:
Dilute sulphuric acid and dilute hydrochloric acid (using barium chloride solution)
Which property of sulphuric acid is shown by the reaction of the concentrated sulphuric acid with carbon?
Choose the current answer from the options given below :
The catalyst used for the oxidation of SO2 to SO3 in contact process is
What do you observe when barium chloride solution is added to dilute sulphuric acid ?
Which concentrated acid oxidizes sulphur directly to sulphuric acid ? Write the equation for the same. What is the name of the process by which sulphuric acid is manufactured ? Name the catalyst used in the process.
Complete the following sentence, choosing the correct word from the given options.
"Concentrated sulphuric acid is used in the laboratory preparation of nitric acid and hydrochloric acid because it is ____________ in comparison to these two acids."
Write the equation for the laboratory preparation of the following salts, using sulphuric acid.
Lead sulphate from lead nitrate.
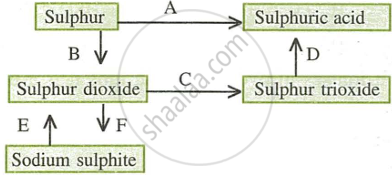
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
State one appropriate observation of the following:
Dehydration of concentrated sulphuric acid with sugar crystals.
Give one equation to show the following property of sulphuric acid:
As a non-volatile acid
