Advertisements
Advertisements
प्रश्न
Which property of sulphuric acid is shown by the reaction of the concentrated sulphuric acid with carbon?
State the property of sulphuric acid shown by the reaction of conc. sulphuric acid when heated with carbon.
Advertisements
उत्तर
It behaves as an oxidising agent and oxidises carbon to carbon dioxide.
\[\ce{C + 2H2SO4 -> CO2\uparrow + 2H2O + 2SO2\uparrow}\]
APPEARS IN
संबंधित प्रश्न
State one relevant observation for given reactions:
Addition of ethyl alcohol to acetic acid in the presence of concentrated Sulphuric acid
A, B, C, and D summarize the properties of sulphuric acid depending on whether it is dilute or concentrated.
A = Typical acid property
B = Non volatile acid
C = Oxidizing agent
D = Dehydrating agent
Choose the property (A, B, C or D) depending on which is relevant to each of the following
1) Preparation of Hydrogen chloride gas
2) Preparation of Copper sulphate from copper oxide.
3) An action of conc. Sulphuric acid on Sulphur.
Give balanced chemical equations for the action of sulphuric acid on the following:
Sulphur
Quicklime is not used to dry HCl gas because _____.
Give one equation to show the following properties of sulphuric acid:
Dehydrating property
Give reason for the following:
When concentrated sulphuric acid is added to blue crystalline copper sulphate, it turns powdery white.
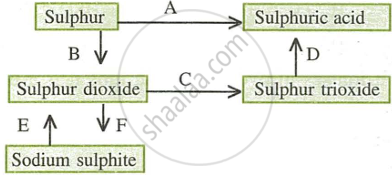
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
Write the equation of the following reaction:
Sulphur is heated with concentrated sulphuric acid.
What would you observe in the following case:
Sugar crystals are added to a hard glass test tube containing concentrated sulphuric acid.
Identify the gas evolved and give the chemical test in the following case:
Dilute hydrochloric acid reacts with sodium sulphite.
