Advertisements
Advertisements
प्रश्न
How is ethyl amine prepared from methyl iodide?
Advertisements
उत्तर
Methyl iodide reacts with alcoholic solution of potassium cyanide (KCN) to give ethane nitrile (or methyl cyanide) as the major product with a small amount of potassium iodide.

Ethane nitrile (or methyl cyanide) on reduction with sodium and alcohol (Mendius reaction) forms ethyl amine.

APPEARS IN
संबंधित प्रश्न
An aromatic compound 'A' of molecular formula C7H7ON undergoes a series of reactions as shown below. Write the structures of A, B, C, D and E in the following reactions :

Write a short note on the following:
Hoffmann’s bromamide reaction
Give the structure of A, B and C in the following reaction:
\[\ce{CH3CH2I ->[NaCN] A ->[OH-][Partial hydrolysis] B ->[NaOH + Br2] C}\]
Give the structure of A, B and C in the following reaction:
\[\ce{C6H5N2Cl ->[CuCN] A ->[H2O/H+] B ->[NH3][\Delta] C}\]
Give the structures of A, B and C in the following reaction:
\[\ce{CH3COOH ->[NH3][\Delta] A ->[NaOBr] B ->[NaNO2/HCl] C}\]
Give the structures of A, B and C in the following reaction:
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[HNO2][273 K] B ->[C6H5OH] C}\]
Why cannot aromatic primary amines be prepared by Gabriel phthalimide synthesis?
Write the reaction of aromatic primary amine with nitrous acid.
Write the reaction of aliphatic primary amine with nitrous acid.
Arrange the following in the increasing order of their pKb values:
C6H5NH2, C2H5NH2, C6H5NHCH3
Give the structures of A, B and C in the following reactions :
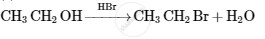
Answer in one sentence.
Predict the product of the following reaction.
\[\ce{Nitrobenzene ->[Sn/conc.HCl]?}\]
Answer the following
Write a reaction to convert acetic acid into methylamine.
Write the order of reactivity of alkyl halides with ammonia.
Explain Hoffmann’s exhaustive alkylation with suitable reactions.
Write reactions for the preparation of ethanamine using Gabriel phthalimide synthesis.
Explain the following reaction with a suitable example.
Hofmann elimination reaction
The end product C of the following reaction is
\[\ce{C2H5NH2 ->[HNO2] A ->[PCl5] B ->[NH3][Alcohol] C}\]
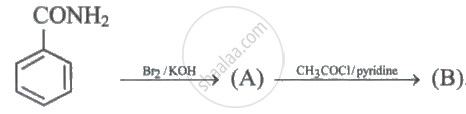
Identify the major product (B).
____________ can be prepared exclusively by Gabriel phthalimide synthesis.
What product is formed when \[\ce{R - C ≡ N}\] is hydrolysed?
\[\ce{CH3-CN ->[Na/C2H5OH]}\]
The product formed is ____________.
The reduction of alkyl cyanide with sodium and ethanol to give primary amines is, ____________.
Identify the INCORRECT statement regarding Hofmann bromamide reaction.
Which of the following amines cannot be prepared by Gabriel phthalimide synthesis?
Identify product B in the following reaction.
\[\ce{Aniline ->[NaNO2][HCl] A ->[KI] B}\]
Which of the following reagents is used in Hofmann's elimination reaction of amines?
Which of the following does NOT give carbylamine test?
Nitro compounds are reduced by iron scrap and hydrochloric acid to yield one of the following compounds:
For producing amines, the reaction of nitro compounds with iron scrap is preferred because:
Amongst the following, the strongest base in aqueous medium is ______.
Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
The source of nitrogen in Gabriel synthesis of amines is ______.
Best method for preparing primary amines from alkyl halides without changing the number of carbon atoms in the chain is ______.
Which of the following compounds is the weakest Brönsted base?
Which of the following amines can be prepared by Gabriel synthesis.
(i) Isobutyl amine
(ii) 2-Phenylethylamine
(iii) N-methylbenzylamine
(iv) Aniline
What is the product when \[\ce{C6H5CH2NH2}\] reacts with \[\ce{HNO2}\]?
How will you carry out the following conversions?

How will you carry out the following conversions?

Match the reactions given in Column I with the statements given in Column II.
| Column I | Column II | ||
| (i) | Ammonolysis | (a) | Amine with lesser number of carbon atoms |
| (ii) | Gabriel phthalimide synthesis | (b) | Detection test for primary amines. |
| (iii) | Hoffmann Bromamide reaction | (c) | Reaction of phthalimide with \[\ce{KOH}\] and \[\ce{R-X}\] |
| (iv) | Carbylamine reaction | (d) | Reaction of alkylhalides with \[\ce{NH3}\] |
Account for the following:
Aniline cannot be prepared by the ammonolysis of chlorobenzene under normal conditions.
A primary amine is formed by an amide on treatment with bromine and alkali. The primary amine has
C6H5CONHCH3 can be converted into C6H5CH2NHCH3 by:-
When primary amines are treated with HCl, the product obtained is which of the following?
Which of the following compound gives pink colour on reaction with phthalic anhydride in cone. H2SO4 followed by treatment with NaOH?
Identify the product ‘C’ in the following reaction.
\[\ce{Aniline ->[(CH3CH)2O][Pyridine] A ->[Br2][CH3COOH] B ->[H^+ or OH^-] C}\]
Identify the compo ds A and B in the following reactions:
\[\ce{A ->[Nitrating mixture] B ->[(i) Sn/cone. HCI][(ii) NaOH] Aniline}\]
Write a short note on the following:
Ammonolysis
Write a short note on the following:
Ammonolysis
Write short note on the following.
Ammonolysis.
Write short notes on the following:
Ammonolysis
