Topics
Periodic Table, Periodic Properties and Variations of Properties
- The Modern Periodic Table
- Salient Features of the Modern Periodic Table
- Periodicity
- Shells and Valency
- Periodic Properties
- Atomic Size
- Metallic Character
- Non-metallic Character
- Ionisation Potential (Ionisation Energy)
- Electron Affinity
- Electronegativity
- Summary Periodic Properties
- Atomic Number and Mass Number
- Comparison of Alkali Metals and Halogens
Chemical Bonding
- Concept of Chemical Bonding
- Ionic or Electrovalent Bond
- Conditions for the Formation of an Electrovalent (or ionic) Bond
- Structures of Some Electrovalent Compounds
- The Covalent Bond
- Types of Covalent Bonds
- Non-Polar and Polar Covalent Compounds
- Formation of Covalent Bond
- Properties and Comparison of Electrovalent and Covalent Compounds
- Effect of Electricity on Electrovalent and Covalent Compounds
- Coordinate Bond
Acids, Bases and Salts
Acids and Bases
- Basics of Acids, Bases, and Salts
- Acids
- Classification of Acids
- Preparation of Acids
- Properties of Acids > Physical Properties
- Properties of Acids > Chemical Properties
- Uses of Acids
- Bases (Alkalis)
- Classification of Bases (Alkalis)
- Preparation of Bases
- Properties of Bases > Physical Properties
- Properties of Bases > Chemical Properties
- Uses of Bases
- Test for Acidity and Alkalinity
- Importance of pH in Everyday Life
Salts and their Preparations
Properties of Salts
- General Properties of Salts
- Differences Between Drying Agent and Dehydrating Agent
Analytical Chemistry
- Chemical Analysis
- Colours of the Salts and Their Solutions
- Action of Sodium Hydroxide Solution on Certain Metallic Salt Solutions
- Action of Ammonium Hydroxide on Certain Salt Solutions
- Action of Alkalis on Certain Metals
- Action of Alkalis on Metal Oxides
Mole Concept and Stoichiometry
Gay-Lussac's Law and Avogadro's Law
Relative Atomic Mass, Relative Molecular Mass and Mole Concept
Percentage Composition, Empirical and Molecular Formulae
Calculations Based on Chemical Equations
- Chemical Equations
- Problems Based On Reacting Weights
- Problems Based on Mass-Volume Relationship
- Challenging Problems Beyond the Syllabus
Electrolysis
- Electrolysis and Conductivity of Substances
- Metallic Conductors and Electrolytic Conductors
- Strong Electrolytes vs Weak Electrolytes
- Acids, Bases and Salts as Electrolytes
- Anode vs Cathode
- Cations vs Anions
- Oxidation
- Chemical Properties of Carbon Compounds > Reduction
- Theory of Electrolytic Dissociation
- Conduction in Compounds
- Characteristics of Electrolysis
- Electrolytic Dissociation
- Ionisation
- Comparison of Ionisation and Dissociation
- Electrochemical Series (Electromotive Series)
- Preferential or Selective Discharge of Ions at Electrodes
- Examples of Electrolysis
- Applications of Electrolysis > Electroplating
- Applications of Electrolysis > Electrolytic Refining of Metals
- Applications of Electrolysis > Electrometallurgy
Metallurgy
Metals - Occurence
- Earth and Elements
- Metals
- Non-Metals
- Occurrence of Metals
- Common Ores of Aluminium, Iron and Zinc
Stages Involved in the Extraction of Metals
Extraction of Aluminium
- Aluminium
- Extraction of Aluminium
- Electrolytic Reduction of Refused Alumina
- Refining of Aluminium
Alloys
- Alloy
- Purpose of Making Alloys
- Reasons for Alloying
- Methods of Making Alloys
- Alloys as Solid Solutions
Study of Compounds A. Hydrogen Chloride
- Hydrogen Chloride
- General Preparation of Hydrogen Chloride Gas
- Recognition and Identification of Gases
- Physical Properties of Hydrogen Chloride Gas
- Chemical Properties of Hydrogen Chloride Gas
- Hydrochloric Acid
- Laboratory Method of Preparation of Hydrochloric Acid
- Properties of Hydrochloric Acid > Physical Properties
- Properties of Hydrochloric Acid > Chemical Properties
- Uses of Hydrochloric Acid
- Tests for Hydrogen Chloride and Hydrochloric Acid
Study of Compounds B. Ammonia
- Ammonia
- Preparation of Ammonia Gas
- Recognition and Identification of Gases
- Preparation of Aqueous Ammonia
- Manufacture of Ammonia (Haber's Process)
- Physical Properties of Ammonia
- Chemical Properties of Ammonia
- Tests for Ammonia Gas and Ammonium Ion
- Uses of Ammonia
Study of Compounds C. Nitric Acid
- Nitric Acid
- Laboratory Preparation of Nitric Acid
- Manufacture of Nitric Acid
- Physical Properties of Nitric Acid
- Chemical Properties of Nitric Acid
- Uses of Nitric Acid
- Tests for Nitric Acid and Nitrates
- Effects of Heat on Nitrates
Study of Compounds D. Sulphuric Acid
- Sulphuric Acid
- Manufacture of Sulphuric Acid
- Physical Properties of Sulphuric Acid
- Chemical Properties of Sulphuric Acid
- Uses of Sulphuric Acid
- Tests for Sulphuric Acid and Sulphates
Organic Chemistry
Organic Compounds
- Organic Chemistry
- Organic Compounds
- Organic Compounds vs Inorganic Compounds
- Carbon: A Versatile Element
- Types of Organic Compounds
- Hydrocarbons
- Cyclic or Closed Chain or Ring Chain Compounds
- Structure of Compounds
- Alkyl Group
- Identification of Functional Groups
- Homologous Series
- Nomenclature
- Nomenclature of Organic Compounds
- Structural Formula from IUPAC Name
- Isomerism
- Types of Structural Isomerism
Hydrocarbons : Alkanes
- Alkanes
- Isomerism in Alkenes
- Occurrence of Methane and Ethane
- Structure of Methane
- Laboratory Preparation of Methane
- Laboratory Preparation of Ethane
- Other Methods of Preparation of Methane and Ethane
- Physical Properties of Methane and Ethane
- Chemical Properties of Methane and Ethane
- Uses of Methane and Ethane
Hydrocarbons: Alkenes
- Alkenes
- Ethene (Ethylene)
- Preparation of Ethene (Ethylene)
- Physical Properties of Alkenes
- Chemical Properties of Alkenes
- Uses of Ethene
Hydrocarbons: Alkynes
- Alkynes
- Ethyne
- Physical Properties of Ethyne
- Chemical Properties of Ethyne
- Uses of Ethyne
- Chemical Tests to distinguish between Alkanes, Alkenes and Alkynes
Alcohols
Carboxylic Acids
Practical Work
- Recognition and Identification of Gases
- Action of Heat
- Identifying Substances by Colour, Smell, State & Heat Effect
- Distinction between Colourless Solutions of Dilute Acids and Alkalis
- Distinguish between Black Copper Oxide and Black Manganese Dioxide
- Flame Test
- Indicators
Estimated time: 12 minutes
- Key Points: Chemical Properties of Alkenes
CBSE: Class 12
Maharashtra State Board: Class 12
Maharashtra State Board: Class 12
Key Points: Chemical Properties of Alkenes
Alkenes undergo mainly electrophilic addition reactions due to the π bond (electron-rich site).
| Reaction | Example |
|---|---|
| Addition of hydrogen (Hydrogenation) |
\[\ce{H2C = CH2 ->[H2/Ni, Pt or Pd][523-573K] H3C - CH3}\] |
| Addition of halogen | \[\begin{array}{cc} \phantom{}\ce{H3CCH = CH2 + Cl — Cl ->[CCl4] CH3CH - CH2}\phantom{}\\ \phantom{....................................................}|\phantom{.........}|\phantom{}\\ \phantom{.....................................................}\ce{Cl}\phantom{.......}\ce{Cl}\phantom{} \end{array}\] |
| Addition of HX (Markovnikov's rule) | \[\begin{array}{cc} \phantom{..............................................................}\ce{Br}\phantom{}\\ \phantom{............................................................}|\phantom{}\\ \phantom{}\ce{\underset{(For unsymmetrical allkene-Markownikoff’s rule)}{H3CCH = CH2 + HBr} -> H3C - CH - CH3}\phantom{} \end{array}\] |
| Addition of HBr (Anti-Markovnikov / Kharasch effect) |
\[\begin{array}{cc} |
| Hydration (addition of H₂SO₄/H₂O) | \[\begin{array}{cc} \ce{O}\phantom{..}\\ ||\phantom{..}\\ \phantom{}\ce{CH2 = CH2 + H - O - S - O - H -> C2H5HSO4}\phantom{}\\ ||\phantom{..}\\ \ce{O}\phantom{..} \end{array}\] |
| Oxidation (KMnO₄/H⁺) | \[\begin{array}{cc} \phantom{..........................}\ce{O}\phantom{}\\ \phantom{..........................}||\\ \phantom{}\ce{H3C — CH = CH2 ->[{[O]}][KMnO4, {[H^{+}]}] H3C - C - OH + CO2 + H2O}\phantom{} \end{array}\] |
| Hydroxylation | \[\begin{array}{cc} \phantom{}\ce{H2C = CH2 + H2O + [O] ->[Dil.KMnO4][273K] CH2 - CH2}\\ \phantom{.....................................................}|\phantom{..........}|\phantom{}\\ \phantom{........................................................}\ce{OH}\phantom{.....}\ce{OH}\phantom{} \end{array}\] |
| Ozonolysis | \[\begin{array}{cc} \phantom{.....}\ce{H3C}\phantom{....................................}\ce{H3C}\phantom{............................}\\ \phantom{.....}\backslash\phantom{.........................................}\backslash\phantom{.....................}\\ \phantom{..........}\ce{C = CH2 + O3 ->[Zn/H2O] \phantom{.......}C = O + HCHO}\phantom{}\\ \phantom{......}/\phantom{..........................................}/\phantom{.....................}\\ \phantom{...............}\ce{H3C}\phantom{......................................}\ce{H3C}\phantom{......................................} \end{array}\] |
| Polymerisation | |
| Hydroboration-oxidation | \[\begin{array}{cc} \phantom{...............}\ce{H}\phantom{....}\ce{H}\phantom{.............................................}\ce{H}\phantom{....}\ce{H}\phantom{..........................}\ce{H}\phantom{....}\ce{H}\phantom{............................}\\ \phantom{.............}|\phantom{......}|\phantom{..............................................}|\phantom{......}|\phantom{............................}|\phantom{......}|\phantom{..........................}\\ \phantom{}\ce{6(H - C = C - H) + (BH3)2 ->[THF] 2(H - C - C)3 - B ->[H2O2][OH^Θ] H - C - C - H + B(OH)3}\phantom{}\\ \phantom{..........................................}|\phantom{......}|\phantom{............................}|\phantom{......}|\\ \phantom{..............................................}\ce{H}\phantom{.....}\ce{H}\phantom{..........................}\ce{H}\phantom{.....}\ce{OH}\phantom{.} \end{array}\] |
CISCE: Class 10
Example: Ethene
Ethene undergoes addition reactions:
- With hydrogen → ethane
- With halogens → dihaloalkanes
- With steam (acid catalyst) → ethanol
Forms poly(ethene) (polythene) through polymerisation.
Burns with a sooty flame.
On oxidation with KMnO₄, forms ethylene glycol.
Related QuestionsVIEW ALL [18]
Which of the following alkenes on ozonolysis give a mixture of ketones only?
| (i) | CH3 – CH = CH – CH3 |
| (ii) | \[\begin{array}{cc} \ce{CH3 - C - CH = CH2}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
| (iii) | 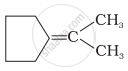 |
| (iv) | \[\begin{array}{cc} \phantom{...................}\ce{CH3}\\ \phantom{..............}/\\ \ce{(CH3)2 C = C}\\ \phantom{..............}\backslash\\ \phantom{...................}\ce{CH3} \end{array}\] |
