Advertisements
Advertisements
Question
Write a chemical equation for combustion reaction of the following hydrocarbon:
Butane
Advertisements
Solution
\[\ce{\underset{Butane}{2C4H_{10(g)}} + 13O_{2(g)} -> 8CO_{2(g)} + 10H2O_{(g)} + Heat}\]
RELATED QUESTIONS
Write IUPAC name of the product obtained by the ozonolysis of the following compound.
Pent-2-ene
An alkene ‘A’ contains three C – C, eight C – H σ bonds and one C – C π bond. ‘A’ on ozonolysis gives two moles of an aldehyde of molar mass 44 u. Write IUPAC name of ‘A’.
Propanal and pentan-3-one are the ozonolysis products of an alkene? What is the structural formula of the alkene?
Write a chemical equation for combustion reaction of the following hydrocarbon:
Pentene
Write a chemical equation for combustion reaction of the following hydrocarbon:
Toluene
Arrange the halogens F2, Cl2, Br2, I2, in order of their increasing reactivity with alkanes.
The addition of HBr to 1-butene gives a mixture of products A, B and C
| (A) | 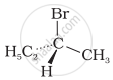 |
| (B) |  |
| (C) | CH3 – CH2 – CH2 – CH2 – Br |
The mixture consists of:
Arrange the following hydrogen halides in order of their decreasing reactivity with propene.
Which of the following alkenes on ozonolysis give a mixture of ketones only?
| (i) | CH3 – CH = CH – CH3 |
| (ii) | \[\begin{array}{cc} \ce{CH3 - C - CH = CH2}\\ |\phantom{.......}\\ \ce{CH3}\phantom{....} \end{array}\] |
| (iii) | 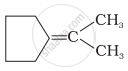 |
| (iv) | \[\begin{array}{cc} \phantom{...................}\ce{CH3}\\ \phantom{..............}/\\ \ce{(CH3)2 C = C}\\ \phantom{..............}\backslash\\ \phantom{...................}\ce{CH3} \end{array}\] |
In the presence of peroxide addition of HBr to propene takes place according to anti Markovnikov’s rule but peroxide effect is not seen in the case of HCl and HI. Explain.
Which of the following reagent is used for the following reaction?
\[\ce{CH3CH2CH3 ->[?] CH3CH2CHO}\]

Select schemes A, B, C out of
(I) acid catalysed hydration
(II) HBO
(III) oxymercuration-demercuration
What would be the main product when propene reacts with HBr?
Propene, \[\ce{CH3 - CH = CH2}\] can be converted to 1-propanol by oxidation. Which set of reagents among the following is ideal to effect the conversion ______.
3-Methyl-pent-2-ene on reaction with HBr in presence of peroxide forms an addition product. The number of possible stereoisomers for the product is ______.
An alkene ‘A’ contains three C – C, eight C – H σ bonds and one C – C π bond. ‘A’ on ozonolysis gives two moles of an aldehyde of molar mass 44 u. Write the IUPAC name of ‘A’.
Identify the correct reagents that would bring about the following transformation.