Advertisements
Advertisements
Question
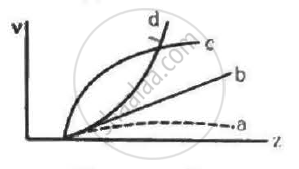
Frequencies of Kα X-rays of different materials are measured. Which one of the graphs in the figure may represent the relation between the frequency v and the atomic number Z ?
Advertisements
Solution
Using Moseley's Law,
`sqrt(v) = a(Z - b)`,
where v = frequency of Kα X-ray
Z = atomic number
`therefore v = a^2(Z - b)^2`
⇒ `(Z - b)^2 = v/a^2`
This is the equation of a parabola with some intercept on the axis, representing atomic number (Z). Hence, curve d represent this relation correctly.
APPEARS IN
RELATED QUESTIONS
What role dose infra-red radiation play in physical therapy?
Name the electromagnetic radiations used for (a) water purification, and (b) eye surgery.
State the approximate range of wavelength associated with visible light.
Name the radiations of wavelength just shorter than 4 × 10-7 m.
Name the waves used for taking photographs in dark.
Can X-rays be used for photoelectric effect?
The energy of a photon of a characteristic X-ray from a Coolidge tube comes from
The Kβ X-rays from certain elements are given below. Draw a Moseley-type plot of √v versus Z for Kβ radiation.
| Element | Ne | P | Ca | Mn | Zn | Br |
| Energy (keV) | 0.858 | 2.14 | 4.02 | 6.51 | 9.57 | 13.3 |
Find the maximum potential difference which may be applied across an X-ray tube with tungsten target without emitting any characteristic K or L X-ray. The energy levels of the tungsten atom with an electron knocked out are as follows.
| Cell containing vacancy | K | L | M |
| Energy in keV | 69.5 | 11.3 | 2.3 |
The Kα X-rays of aluminium (Z = 13) and zinc (Z = 30) have wavelengths 887 pm and 146 pm respectively. Use Moseley's law √v = a(Z − b) to find the wavelengths of the Kα X-ray of iron (Z = 26).
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
The short-wavelength limit shifts by 26 pm when the operating voltage in an X-ray tube is increased to 1.5 times the original value. What was the original value of the operating voltage?
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
The electron beam in a colour TV is accelerated through 32 kV and then strikes the screen. What is the wavelength of the most energetic X-ray photon?
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
| Gamma rays | D | C | Visible light | B | A |
The above table shows different parts of the electromagnetic spectrum.
(a) Identify the parts of the spectrum marked as A, B, C and D.
(b) Which of the radiations A or B has the higher frequency?
(c) State two properties which are common to all parts of the electromagnetic spectrum.
(d) Name one source of each of the radiation of electromagnetic spectrum.
(e) Name one detector for each of the radiation.
(f) Name one use of each of the radiation.
Give one use of electromagnetic radiation in Ultraviolet radiation.
Calculate the wavelength of a microwave of a frequency of 8.0 GHz.
If the Earth did not have atmosphere, would its average surface temperature be higher or lower than what it is now? Explain.
An e.m. wave exerts pressure on the surface on which it is incident. Justify.
Which of the following is a tool used for separating the different color wavelengths from each other?
Light of wavelength `3500A` is incident on two metals A and B whose work functions are 3.2 eV and 1.9 eV respectively. Which metal will emit photoelectrons?
