Advertisements
Advertisements
प्रश्न
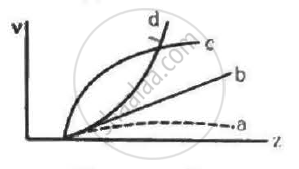
Frequencies of Kα X-rays of different materials are measured. Which one of the graphs in the figure may represent the relation between the frequency v and the atomic number Z ?
Advertisements
उत्तर
Using Moseley's Law,
`sqrt(v) = a(Z - b)`,
where v = frequency of Kα X-ray
Z = atomic number
`therefore v = a^2(Z - b)^2`
⇒ `(Z - b)^2 = v/a^2`
This is the equation of a parabola with some intercept on the axis, representing atomic number (Z). Hence, curve d represent this relation correctly.
APPEARS IN
संबंधित प्रश्न
Why are microwaves considered suitable for radar systems used in aircraft navigation?
If the earth did not have atmosphere, would its average surface temperature be higher or lower than what it is now? Explain.
Give a reason for the following:
It is necessary to use satellites for long-distance TV transmission. Why?
A wave has a wavelength of 10-3 nm. Name the wave.
What is the range of the wavelength of the following electromagnetic waves?
(A) Radio waves.
Name the region beyond the violet end of the spectrum called.
What do you understand by the invisible spectrum?
Can X-rays be used for photoelectric effect?
X-ray from a Coolidge tube is incident on a thin aluminium foil. The intensity of the X-ray transmitted by the foil is found to be I0. The heating current is increased to increase the temperature of the filament. The intensity of the X-ray transmitted by the foil will be
(a) zero
(b) < I0
(c) I0
(d) > I0
The Kα X-rays of aluminium (Z = 13) and zinc (Z = 30) have wavelengths 887 pm and 146 pm respectively. Use Moseley's law √v = a(Z − b) to find the wavelengths of the Kα X-ray of iron (Z = 26).
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
If the operating potential in an X-ray tube is increased by 1%, by what percentage does the cutoff wavelength decrease?
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
An X-ray tube operates at 40 kV. Suppose the electron converts 70% of its energy into a photon at each collision. Find the lowest there wavelengths emitted from the tube. Neglect the energy imparted to the atom with which the electron collides.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Name the scientist who discovered Infra-red waves
State the name and the range of wavelength of the invisible electromagnetic waves beyond the red end of the visible spectrum.
Name the radiation which can be detected by thermopile.
Name any two electromagnetic waves which have a frequency higher than that of violet light. State one use of each.
How will you investigate the existence of the radiation beyond the red and violet extremes of the spectrum?
Microwaves are electromagnetic waves with frequency in the range of.
The electric field intensity produced by the radiations coming from 100 W bulb at a 3 m distance is E. The electric field intensity produced by the radiations coming from 50 W bulb at the same distance is ______.
