Advertisements
Advertisements
प्रश्न
An X-ray tube operates at 40 kV. Suppose the electron converts 70% of its energy into a photon at each collision. Find the lowest there wavelengths emitted from the tube. Neglect the energy imparted to the atom with which the electron collides.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Advertisements
उत्तर
Given:-
Potential of the X-ray tube, V = 40 kV = 40 × 103 V
Energy = 40 × 103 eV
Energy utilised by the electron is given by `E = 70/100 xx 40 xx 10^3 = 28 xx 10^3 "eV"`
Wavelength (`lambda`) is given by
`lambda = (hc)/E`
Here,
h = Planck's constant
c = Speed of light
E = Energy of the electron
`therefore lambda = (hc)/E`
`⇒ lambda = (1242 "eV" - "nm")/(28 xx 10^3 "eV")`
`⇒ lambda = (1242 xx 10^-9 "eV")/(28 xx 10^3 "eV")`
`⇒ lambda = 44.35 xx 10^-12`
`⇒ lambda = 44.35 "pm"`
For the second wavelength,
E = 70% (Leftover energy)
`= 70/100 xx (40-28)10^3`
`= 70/100 xx 12 xx 10^3`
`= 84 xx 10^2 "eV"`
And
`lambda = (hc)/E = 1242/(8.4 xx 10^3)`
`= 147.86 xx 10^-3 "nm"`
`= 147.86 "pm" = 148 "pm"`
For the third wavelength,
`E = 70/100(12 - 8.4) xx 10^3`
`= 7 xx 3.6 xx 10^2 = 25.2 xx 10^2 "eV"`
And ,
`lambda = (hc)/E = 1242/(25.2 xx 10^2)`
`= 49.2857 xx 10^-2`
`= 493 "pm"`
APPEARS IN
संबंधित प्रश्न
How are infrared waves produced?
To which part of the electromagnetic spectrum does a wave of frequency 5 × 1019 Hz belong?
Given below are some famous numbers associated with electromagnetic radiations in different contexts in physics. State the part of the electromagnetic spectrum to which each belongs.
(a) 21 cm (wavelength emitted by atomic hydrogen in interstellar space).
(b) 1057 MHz (frequency of radiation arising from two close energy levels in hydrogen; known as Lamb shift).
(c) 2.7 K [temperature associated with the isotropic radiation filling all space-thought to be a relic of the ‘big-bang’ origin of the universe].
(d) 5890 Å - 5896 Å [double lines of sodium]
(e) 14.4 keV [energy of a particular transition in 57Fe nucleus associated with a famous high resolution spectroscopic method (Mössbauer spectroscopy)].
(a) Give a list of at least five radiations, in order of their increasing frequencies, which make up the complete electromagnetic spectrum.
(b) Which of the radiation mentioned by you in part (a) has the highest penetrating power.
State the approximate range of wavelength associated with the ultraviolet rays.
The wavelength of X-rays is 0.01 Å. Calculate its frequency. State the assumption made, if any.
Is it possible that in a Coolidge tube characteristic Lα X-rays are emitted but not Kα X-rays?
An X-ray beam can be deflected
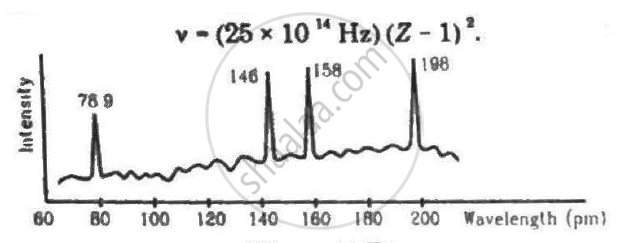
Continuous X-rays are made to strike a tissue paper soaked with polluted water. The incoming X-rays excite the atoms of the sample by knocking out the electrons from the inner shells. Characteristic X-rays are analysed and the intensity is plotted against the wavelength. Assuming that only Kα intensities are detected, list the elements present in the sample from the plot. Use Moseley's equation v − (25 × 1014Hz)(Z − 1)2.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
| Gamma rays | D | C | Visible light | B | A |
The above table shows different parts of the electromagnetic spectrum.
(a) Identify the parts of the spectrum marked as A, B, C and D.
(b) Which of the radiations A or B has the higher frequency?
(c) State two properties which are common to all parts of the electromagnetic spectrum.
(d) Name one source of each of the radiation of electromagnetic spectrum.
(e) Name one detector for each of the radiation.
(f) Name one use of each of the radiation.
Name two sources of ultraviolet radiation.
Name the radiations used for the detection of fracture in bones.
Answer briefly.
Give two uses of ultraviolet rays.
An e.m. wave exerts pressure on the surface on which it is incident. Justify.
The ratio of contributions made by the electric field and magnetic field components to the intensity of an EM wave is ______.
Arrange the following electromagnetic radiation in the ascending order of their frequencies:
X-rays, microwaves, gamma rays, radio waves
Write two uses of the following radiation.
X-rays
In uranium (Z = 92) the K absorption edge is 0.107 Å and the Kα line is 0.126 Å, and the wavelength of the L absorption edge is ______.
In an atom X, electrons absorb the energy from an external source. This energy “excites” the electrons from a lower-energy level to a higher-energy level around the nucleus of the atom. When electrons return to the ground state, they emit photons.
The figure below is the energy level diagram of atom X with three energy levels, E1 = 0.00eV, E2 = 1.78eV and E3 = 2.95eV. The ground state is considered 0 eV for reference. The transition of electrons takes place between levels E1 and E2.

- What wavelength of radiation is needed to excite the atom to energy level E2 from E1?
- Suppose the external source has a power of 100 W. What would be the rate of photon emission?
