Advertisements
Advertisements
Question
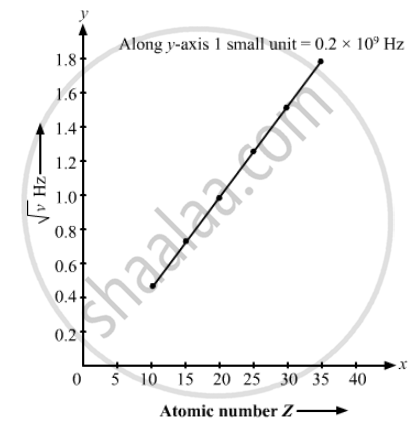
The Kβ X-rays from certain elements are given below. Draw a Moseley-type plot of √v versus Z for Kβ radiation.
| Element | Ne | P | Ca | Mn | Zn | Br |
| Energy (keV) | 0.858 | 2.14 | 4.02 | 6.51 | 9.57 | 13.3 |
Advertisements
Solution
We can directly get value of v by energy frequency relation.
hv = energy
Here, h = Planck's constant
`v = "Energy (in keV)"/h`
The required graph is as follows :
APPEARS IN
RELATED QUESTIONS
The terminology of different parts of the electromagnetic spectrum is given in the text. Use the formula E = hv (for energy of a quantum of radiation: photon) and obtain the photon energy in units of eV for different parts of the electromagnetic spectrum. In what way are the different scales of photon energies that you obtain related to the sources of electromagnetic radiation?
State Moseley's law
An electromagnetic wave has a frequency of 500 MHz and a wavelength of 60 cm Calculate the velocity of the wave.
Which part of electromagnetic spectrum is used in radar systems?
Mark the correct options.
(a) An atom with a vacancy has smaller energy that a neutral atom.
(b) K X-ray is emitted when a hole makes a jump from the K shell to some other shell.
(c) The wavelength of K X-ray is smaller than the wavelength of L X-ray of the same material.
(d) The wavelength of Kα X-ray is smaller than the wavelength of Kβ X-ray of the same material.
The wavelengths of Kα and Lα X-rays of a material are 21.3 pm and 141 pm respectively. Find the wavelength of Kβ X-ray of the material.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
The distance between the cathode (filament) and the target in an X-ray tube is 1.5 m. If the cutoff wavelength is 30 pm, find the electric field between the cathode and the target.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
-
- Calculate the speed of the wave.
- Name the medium through which it is traveling.
State the name and the range of wavelength of the invisible electromagnetic waves beyond the red end of the visible spectrum.
Define the term "Intensity" in the photon picture of electromagnetic radiation.
Gamma rays and radio waves travel with the same velocity in free space. Distinguish between them in terms of their origin and the main application.
Name any two electromagnetic waves which have a frequency higher than that of violet light. State one use of each.
What are the ultraviolet rays?
Answer briefly.
What are radio waves?
Answer briefly.
What is a carrier wave?
A car is moving towards a high cliff. The car driver sounds a horn of frequency f. The reflected sound heard by the driver has a frequency 2f. If v be the velocity of sound, then the velocity of the car, in the same velocity units, will be:
The fundamental frequency of an open organ pipe is 300 Hz. The first overtone of this pipe has same frequency as first overtone of a closed organ pipe. If speed of sound is 330 m/s, then the length of closed organ pipe is:
Find the photon energy in units of ev for electromagnetic wave if waves length 40 m. Given h = 6.63 × 10–34 J.
What is the speed of radio waves in vacuum?
What happens when an electron collides with a positron?
