Advertisements
Advertisements
Question
The short-wavelength limit shifts by 26 pm when the operating voltage in an X-ray tube is increased to 1.5 times the original value. What was the original value of the operating voltage?
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Advertisements
Solution
Let `lambda` be the initial wavelength, V be the initial potential, `lambda^'`be the new wavelength and V' be the new operating voltage when the operating voltage is increased in the X-ray tube.
Given :-
`lambda^' = lambda - 26 "pm"`
V = 1.5 V
Energy (E) is given by
`E = (hc)/lambda`
`⇒ eV = (hc)/lambda`
Here,
h = Planck's constant
c = Speed of light
`lambda` = Wavelength of light
V = Operating potential
`therefore lambda = (hc)/(eV)`
`⇒ lambdaV = lambda^'V^' ............[∵ lambda ∝ 1/V]`
`⇒ lambda V = (lambda - 26) xx 1.5 V`
`⇒ 0.5lambda = 26 xx 1.5`
`⇒ lambda = 26 xx 3`
`⇒ lambda = 78 "pm"`
Hence, the initial wavelength is `78 xx 10^-12 "m".`
Now, the operating voltage (V) is given by
`V = (hc)/(elambda)`
`⇒ V = (6.63 xx 10^-34 xx 3 xx 10^8)/(1.6 xx 10^-19 xx 78 xx 10^-12)`
`⇒ V = 0.15937 xx 10^5`
`⇒ V = 15.9 "kV"`
APPEARS IN
RELATED QUESTIONS
Give a reason for the following:
Long-distance radio broadcasts use short-wave bands. Why?
Optical and radio telescopes are built on the ground but X-ray astronomy is possible only from satellites orbiting the earth. Why?
If the earth did not have an atmosphere, would its average surface temperature be higher or lower than what it is now?
Name two electromagnetic waves of frequency smaller than that of violet light. State one use of each.
Name the waves produced by the changes in the nucleus of an atom.
Can X-rays be polarised?
Is it possible that in a Coolidge tube characteristic Lα X-rays are emitted but not Kα X-rays?
Moseley's Law for characteristic X-ray is √v = a(Z − b). Here,
The electric current in an X-ray tube (from the target to the filament) operating at 40 kV is 10 mA. Assume that on an average, 1% of the total kinetic energy of the electron hitting hte target are converted into X-rays.
(a) What is the total power emitted as X-rays and (b) how much heat is produced in the target every second?
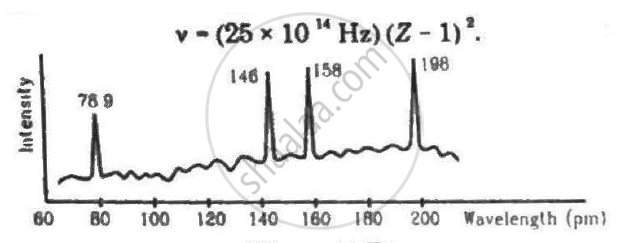
Continuous X-rays are made to strike a tissue paper soaked with polluted water. The incoming X-rays excite the atoms of the sample by knocking out the electrons from the inner shells. Characteristic X-rays are analysed and the intensity is plotted against the wavelength. Assuming that only Kα intensities are detected, list the elements present in the sample from the plot. Use Moseley's equation v − (25 × 1014Hz)(Z − 1)2.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Name the part of the electromagnetic spectrum which is:
Suitable for radar systems used in aircraft navigation.
Gamma rays and radio waves travel with the same velocity in free space. Distinguish between them in terms of their origin and the main application.
Answer briefly.
Why light waves travel in a vacuum whereas sound waves cannot?
Solve the numerical problem.
Calculate the wavelength in nm of an X-ray wave of frequency 2.0 × 1018 Hz.
Calculate the wavelength of a microwave of a frequency of 8.0 GHz.
If the Earth did not have atmosphere, would its average surface temperature be higher or lower than what it is now? Explain.
A bat moving at 10 ms−1 towards a wall sends a sound signal of 8000 Hz towards it. On reflection, it hears a sound of frequency f The value of f in Hz is close to (speed of sound = 320 ms−1)
The fundamental frequency of an open organ pipe is 300 Hz. The first overtone of this pipe has same frequency as first overtone of a closed organ pipe. If speed of sound is 330 m/s, then the length of closed organ pipe is:
Ozone layer above the earth's atmosphere will
The half-value thickness of an absorber is defined as the thickness that will reduce exponentially the intensity of a beam of particles by a factor of 2. The half-value thickness in (µm) for lead assuming X-ray beam of wavelength 20 pm, µ = 50 cm-1 for X-rays in lead at wavelength λ = 20 pm, is ______ µm.
