Advertisements
Advertisements
Question
The energy of a silver atom with a vacancy in K shell is 25.31 keV, in L shell is 3.56 keV and in M shell is 0.530 keV higher than the energy of the atom with no vacancy. Find the frequency of Kα, Kβ and Lα X-rays of silver.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Advertisements
Solution
Given:-
Energy of electron in the K shell, Ek = 25.31 keV
Energy of electron in the L shell, EL = 3.56 keV
Energy of electron in the M shell, EM = 0.530 keV
Let f be the frequency of Kα X-ray and f0 be the frequency of Kβ X-ray.
Let f1 be the frequency of Lα X-rays of silver.
∴ Kα = EK − EL = hf
Here, h = Planck constant
f = frequency of `Kα` X-ray
`f = (E_K - E_L)/h`
`f = ((25.31 xx 3.56))/(6.63 xx 10^-34) xx 1.6 xx 10^-19 xx 10^3`
`f = (21.75 xx 10^3 xx 10^15)/6.67`
`f = 5.25 xx 10^18 "Hz"`
`K_β = E_K - E_M = hf_0`
`⇒ f_0 = (E_K - E_M)/h`
`⇒ f_0 = ((25.31 - 0.53))/(6.67 xx 10^-34) xx 10^3 xx 1.6 xx 10^-19`
`⇒ f_0 = 5.985 xx 10^18 "Hz"`
`K_L = E_L - E_M = hf_1`
`f_1 = (E_L - E_M)/h`
`f_1 = (3.56 - 0.530)/(6.63 xx 10^-34) xx 10^3 xx 1.6 xx 10^-19`
`f_1 = 7.32 xx 10^17 "Hz"`
APPEARS IN
RELATED QUESTIONS
What role dose infra-red radiation play in maintain the Earth’s warmth
A plane electromagnetic wave travels in vacuum along z-direction. What can you say about the directions of its electric and magnetic field vectors? If the frequency of the wave is 30 MHz, what is its wavelength?
The terminology of different parts of the electromagnetic spectrum is given in the text. Use the formula E = hv (for energy of a quantum of radiation: photon) and obtain the photon energy in units of eV for different parts of the electromagnetic spectrum. In what way are the different scales of photon energies that you obtain related to the sources of electromagnetic radiation?
Give a reason for the following:
It is necessary to use satellites for long-distance TV transmission. Why?
Optical and radio telescopes are built on the ground but X-ray astronomy is possible only from satellites orbiting the earth. Why?
What is the range of the wavelength of the following electromagnetic waves?
(a) Visible.
State the approximate range of wavelength associated with visible light.
Name of physical quantity which remains same for microwaves of wavelength 1 mm and UV radiations of 1600 Å in vacuum.
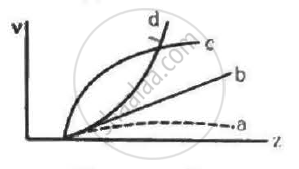
Frequencies of Kα X-rays of different materials are measured. Which one of the graphs in the figure may represent the relation between the frequency v and the atomic number Z ?
Write the range of the wavelength of the following electromagnetic radiations:
(a) Infrared rays
(b) Ultraviolet rays
(c) γ -rays
Write one use of each of the above.
Name the scientist who discovered
X-rays
Name the scientist who discovered radio waves
Name the radiation of the electromagnetic spectrum which is used for the following:
To photograph internal parts of the human body and Give the frequency range
Following QN ∴ 14, the radiation force on the roof will be
Electromagnetic waves with wavelength
- λ1 is used in satellite communication.
- λ2 is used to kill germs in water purifies.
- λ3 is used to detect leakage of oil in underground pipelines.
- λ4 is used to improve visibility in runways during fog and mist conditions.
- Identify and name the part of electromagnetic spectrum to which these radiations belong.
- Arrange these wavelengths in ascending order of their magnitude.
- Write one more application of each.
Which is the correct ascending order of wavelengths?
Identify the part of the electromagnetic spectrum which:
- produces the heating effect.
- is absorbed by the ozone layer in the atmosphere.
- is used for studying crystal structure.
Write any one method of the production of each of the above radiations.
Name one radiation having the wavelength longer than the wavelength of these radiations.
Name the electromagnetic radiation that has been used in obtaining the image below.