Advertisements
Advertisements
Question
Calculate the wavelength of a microwave of a frequency of 8.0 GHz.
Advertisements
Solution
Given: ν = 8 GHz = 8 × 109 Hz, c = 3 × 108 m/s
To find: Wavelength (λ)
Formula: c = νλ
Calculation: From formula,
λ = `"c"/ν`
= `(3xx10^8)/(8xx10^9)`
= 3.75 × 10−2
= 3.75 cm
The wavelength of microwave is 3.75 cm.
APPEARS IN
RELATED QUESTIONS
What role dose infra-red radiation play in maintain the Earth’s warmth
Identify the part of the electromagnetic spectrum which is suitable for radar system used in aircraft navigation.
Use the formula λm T= 0.29 cm K to obtain the characteristic temperature ranges for different parts of the electromagnetic spectrum. What do the numbers that you obtain tell you?
Name the phenomenon which shows the quantum nature of electromagnetic radiation.
What is the range of the wavelength of the following electromagnetic waves?
(a) Ultraviolet
What is the range of the wavelength of the following electromagnetic waves?
(a) Micro waves .
Name two electromagnetic waves of frequency smaller than that of violet light. State one use of each.
Give one use of ultraviolet radiation.
Can Lα X-ray of one material have shorter wavelength than Kα X-ray of another?
Why is exposure to X-rays injurious to health but not exposure to visible light, when both are electromagnetic waves?
Consider a photon of a continuous X-ray coming from a Coolidge tube. Its energy comes from
X-ray from a Coolidge tube is incident on a thin aluminium foil. The intensity of the X-ray transmitted by the foil is found to be I0. The heating current is increased to increase the temperature of the filament. The intensity of the X-ray transmitted by the foil will be
(a) zero
(b) < I0
(c) I0
(d) > I0
For harder X-rays,
(a) the wavelength is higher
(b) the intensity is higher
(c) the frequency is higher
(d) the photon energy is higher.
Consider a photon of continuous X-ray and a photon of characteristic X-ray of the same wavelength. Which of the following is/are different for the two photons?
The X-ray coming from a Coolidge tube has a cutoff wavelength of 80 pm. Find the kinetic energy of the electrons hitting the target.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
The Kα and Kβ X-rays of molybdenum have wavelengths 0.71 A and 0.63 A respectively. Find the wavelength of Lα X-ray of molybdenum.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
The electron beam in a colour TV is accelerated through 32 kV and then strikes the screen. What is the wavelength of the most energetic X-ray photon?
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
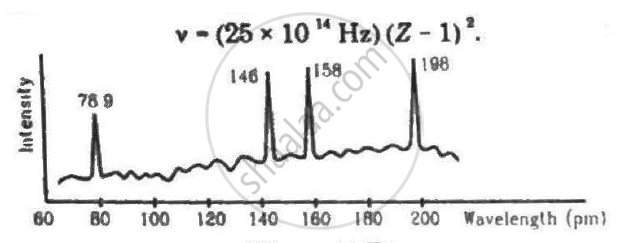
Continuous X-rays are made to strike a tissue paper soaked with polluted water. The incoming X-rays excite the atoms of the sample by knocking out the electrons from the inner shells. Characteristic X-rays are analysed and the intensity is plotted against the wavelength. Assuming that only Kα intensities are detected, list the elements present in the sample from the plot. Use Moseley's equation v − (25 × 1014Hz)(Z − 1)2.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Name three radiations and their wavelength range which are invisible and beyond the violet end of the visible spectrum.
Define the term "Intensity" in the photon picture of electromagnetic radiation.
Answer briefly.
Can we produce a pure electric or magnetic wave in space? Why?
Answer briefly.
Name the most harmful radiation entering the Earth's atmosphere from outer space.
Solve the numerical problem.
Calculate the wavelength in nm of an X-ray wave of frequency 2.0 × 1018 Hz.
For television broadcasting, the frequency employed is normally
The frequency of x-rays, y-rays and ultraviolet rays are respectively a, b and c. Then:-
Find the photon energy in units of ev for electromagnetic wave if waves length 40 m. Given h = 6.63 × 10–34 J.
Electromagnetic waves with wavelength
- λ1 is used in satellite communication.
- λ2 is used to kill germs in water purifies.
- λ3 is used to detect leakage of oil in underground pipelines.
- λ4 is used to improve visibility in runways during fog and mist conditions.
- Identify and name the part of electromagnetic spectrum to which these radiations belong.
- Arrange these wavelengths in ascending order of their magnitude.
- Write one more application of each.
Arrange the following electromagnetic radiation in the ascending order of their frequencies:
X-rays, microwaves, gamma rays, radio waves
What happens when an electron collides with a positron?
