Topics
Units and Measurements
- Quantitative Science
- System of Units
- Derived Quantities and Units
- Rules and Conventions for Writing SI Units and Their Symbols
- Measurement of Length
- Measurement of Mass
- Measurement of Time
- Dimensions and Dimensional Analysis
- Accuracy, Precision and Uncertainty in Measurement
- Errors in Measurements>Systematic Errors
- Errors in Measurements>Random Errors
- Estimation of Errors
- Combination of Errors
- Significant Figures
- Definitions of SI Units and Constants
Mathematical Methods
- Vector Analysis
- Scalar
- Vector
- Vector Operations>Multiplication of a Vector by a Scalar
- Vector Operations>Addition and Subtraction of Vectors
- Vector Operations>Triangle Law for Vector Addition
- Vector Operations>Law of parallelogram of vectors
- Resolution of Vectors
- Multiplication of Vectors>Scalar Product(Dot Product)
- Multiplication of Vectors>Vector Product (Cross Product)
- Concept of Calculus
- Differential Calculus
- Integral Calculus
Motion in a Plane
- Concept of Motion
- Rectilinear Motion
- Displacement
- Path Length
- Average Velocity
- Average Speed
- Instantaneous Velocity
- Instantaneous Speed
- Acceleration in Linear Motion
- Relative Velocity
- Motion in Two Dimensions - Motion in a Plane
- Average and Instantaneous Velocities
- Acceleration in a Plane
- Equations of Motion in a Plane with Constant Acceleration
- Relative Velocity in Two Dimensions
- Projectile Motion
- Uniform Circular Motion (UCM)
- Key Parameters of Circular Motion
- Centripetal Acceleration
- Conical Pendulum
Laws of Motion
- Fundamental Principles of Motion and Mechanics
- Types of Motion
- Aristotle’s Fallacy
- Newton’s Laws of Motion
- Newton's First Law of Motion
- Newton’s Second Law of Motion
- Newton's Third Law of Motion
- Inertial and Non-inertial Frames of Reference
- Types of Forces>Fundamental Forces in Nature
- Types of Forces>Contact and Non-Contact Forces
- Types of Forces>Real and Pseudo Forces
- Types of Forces>Conservative and Non-Conservative Forces
- Types of Forces>Work Done by a Variable Force
- Work Energy Theorem
- Principle of Conservation of Linear Momentum
- Collisions
- Elastic and Inelastic Collisions
- Perfectly Inelastic Collision
- Coefficient of Restitution e
- Expressions for Final Velocities in Elastic Head-On Collision
- Loss of Kinetic Energy in Perfectly Inelastic Head-On Collision
- Collision in Two Dimensions
- Impulse of a Force
- Necessity of Defining Impulse
- Rotational Analogue of a Force: Moment of a Force Or Torque
- Couple and Its Torque
- Proof of Independence of the Axis of Rotation
- Mechanical Equilibrium
- States of Equilibrium
- Centre of Mass>Mathematical Understanding of Centre of Mass
- Centre of Mass>Velocity of Centre of Mass
- Centre of Mass>Acceleration of Centre of Mass
- Centre of Mass>Characteristics of Centre of Mass
- Centre of Gravity
Gravitation
- Concept of Gravitation
- Kepler’s Laws
- Law of Orbit or Kepler's First Law
- Law of Areas or Kepler's Second Law
- Law of Periods or Kepler's Third Law
- Newton's Universal Law of Gravitation
- Measurement of the Gravitational Constant (G)
- Acceleration Due to Gravity (Earth’s Gravitational Acceleration)
- Variation in the Acceleration>Variation in Gravity with Altitude
- Variation in the Acceleration>Variation in Gravity with Depth
- Variation in the Acceleration>Variation in Gravity with Latitude and Rotation of the Earth
- Variation in the Acceleration>Effect of the Shape of the Earth
- Gravitational Potential Energy
- Expression for Gravitational Potential Energy
- Connection of Potential Energy Formula with mgh
- Potential and Potential Difference
- Escape Velocity
- Earth Satellites
- Projection of Satellite
- Weightlessness in a Satellite
- Time Period of Satellite
- Binding Energy of an Orbiting Satellite
Mechanical Properties of Solids
- Understanding Mechanical Properties of Solids
- Elastic Behavior of Solids
- Stress and Strain
- Types of Stress and Corresponding Strain
- Hooke’s Law
- Elastic Modulus>Young’s Modulus
- Elastic Modulus>Bulk Modulus
- Elastic Modulus>Shear Modulus (Modulus of Rigidity)
- Elastic Modulus>Poisson’s Ratio
- Stress-strain Curve
- Strain Energy
- Hardness of Material
- Friction in Solids
- Origin of Friction
- Types of Friction>Static Friction
- Types of Friction>Kinetic Friction
- Types of Friction>Rolling Friction
Thermal Properties of Matter
- Understanding Thermal Properties of Matter
- Temperature and Heat
- Measurement of Temperature
- Absolute Zero and Absolute Temperature
- Ideal Gas Equation
- Thermal Expansion
- Linear Expansion
- Areal Expansion
- Volume Expansion
- Specific Heat Capacity of Solids and Liquids
- Relation Between Coefficient of Expansion
- Specific Heat Capacity of Gas
- Heat Equation
- Thermal Capacity
- Calorimetry
- Change of State
- Analysis of Observation>From Point A to B
- Analysis of Observation>From Point B to D
- Evaporation vs Boiling
- Boiling Point and Pressure
- Sublimation
- Phase Diagram
- Gas and Vapour
- Latent Heat
- Heat Transfer
- Conduction
- Thermal Conductivity
- Coefficient of Thermal Conductivity
- Thermal Resistance
- Applications of Thermal conductivity
- Convection
- Application of Convection
- Free and Forced Convection
- Radiation
- Newton’s Law of Cooling
Sound
- Sound Waves
- Common Properties of All Waves
- Transverse Waves
- Longitudinal Waves
- Mathematical Expression of a Wave
- The Speed of Travelling Waves
- The Speed of Transverse Waves
- The Speed of Longitudinal Waves
- Newton's Formula for Velocity of Sound
- Laplace’s Correction
- Factors Affecting Speed of Sound
- Principle of Superposition of Waves
- Echo
- Reverberation
- Acoustics
- Qualities of Sound
- Doppler Effect
- Source Moving and Listener Stationary
- Listener Approaching a Stationary Source with Velocity
- Both Source and Listener are Moving
- Common Properties between Doppler Effect of Sound and Light
- Major Differences between Doppler Effects of Sound and Light
Optics
- Fundamental Concepts of Light
- Nature of Light
- Ray Optics Or Geometrical Optics
- Cartesian Sign Convention
- Reflection>Reflection from a Plane Surface
- Reflection>Reflection from Curved Mirrors
- Total Internal Reflection
- Refraction of Light
- Applications of Total Internal Reflection
- Refraction at a Spherical Surface and Lenses
- Thin Lenses and Their Combination
- Refraction at a Single Spherical Surface
- Lens Makers' Equation
- Dispersion of Light
- Analysis of Prism
- Thin Prisms
- Some Natural Phenomena Due to Sunlight
- Defects of Lenses
- Optical Instruments
- Simple Microscope or a Reading Glass
- Compound Microscope
- Telescope
Electrostatics
- Concept of Electrostatics
- Electric Charge
- Basic Properties of Electric Charge
- Additive Nature of Charge
- Quantization of Charge
- Conservation of Charge
- Force between Charges
- Coulomb’s Law
- Scalar Form of Coulomb’s Law
- Relative Permittivity or Dielectric Constant
- Definition of Unit Charge from the Coulomb’s Law
- Coulomb's Law in Vector Form
- Principle of Superposition
- Electric Field
- Electric Field Intensity Due to a Point-Charge
- Practical Way of Calculating Electric Field
- Electric Lines of Force
- Electric Flux
- Gauss’s Law
- Electric Dipole
- Couple Acting on an Electric Dipole in a Uniform Electric Field
- Electric Intensity at a Point Due to an Electric Dipole
- Continuous Charge Distribution
Electric Current Through Conductors
- Electric Current and Its Related Concepts
- Electric Current
- Flow of Current Through a Conductor
- Drift Speed
- Ohm's Law
- Limitations of Ohm’s Law
- Electrical Power
- Resistors
- Rheostat
- Resistors in Parallel
- Specific Resistance or Electrical Resistivity
- Variation of Resistance with Temperature
- Electromotive Force (emf)
- Cells in Series
- Cells in Parallel
- Types of Cells
Magnetism
- Concept of Magnetism
- Magnetic Lines of Force
- Bar Magnet and Solenoid Analogy
- Magnetic Field due to a Bar Magnet
- Magnetic Field Due to a Bar Magnet at an Arbitrary Point
- Gauss' Law of Magnetism
- The Earth’s Magnetism
Electromagnetic Waves and Communication System
- Foundations of Electromagnetic Theory
- Concept of Electromagnetic Waves
- Concept of Electromagnetic Waves
- Characteristics of EM Waves
- Electromagnetic Spectrum
- Radio Waves
- Microwaves
- Infrared waves
- Visible Light
- Ultraviolet rays
- X-rays
- Gamma Rays
- Propagation of EM Waves
- Ground (surface) Wave
- Space wave
- Sky wave propagation
- Communication System
- Elements of a Communication System
- Commonly Used Terms in Electronic Communication System
- Modulation
Semiconductors
- Concept of Semiconductors
- Electrical Conduction in Solids
- Band Theory of Solids
- Intrinsic Semiconductor
- Extrinsic Semiconductor
- n-type Semiconductor
- p-type Semiconductor
- Charge neutrality of extrinsic semiconductors
- Diode or p-n Junction
- Basics of Semiconductor Devices
- Applications of Semiconductors and P-n Junction Diode
- Thermistor
- Introduction
- Definition: Specific Heat Capacity
- Formula: Specific Heat Capacity
- Specific Heat Capacity — Data Table
- Formula: Molar Specific Heat Capacity
- Real-Life Applications
- Key Points: Specific Heat Capacity of Solids and Liquids
Introduction
Every substance has a unique ability to absorb or release heat energy. This property is called specific heat capacity. Here's a classic experiment that reveals it:
Take 1 kg of water and 1 kg of paraffin oil. Heat each with the same heater for the same duration. The paraffin's temperature rises roughly twice as much as the water's — even though both received identical heat energy. This tells us that different substances need different amounts of heat to undergo the same temperature change.
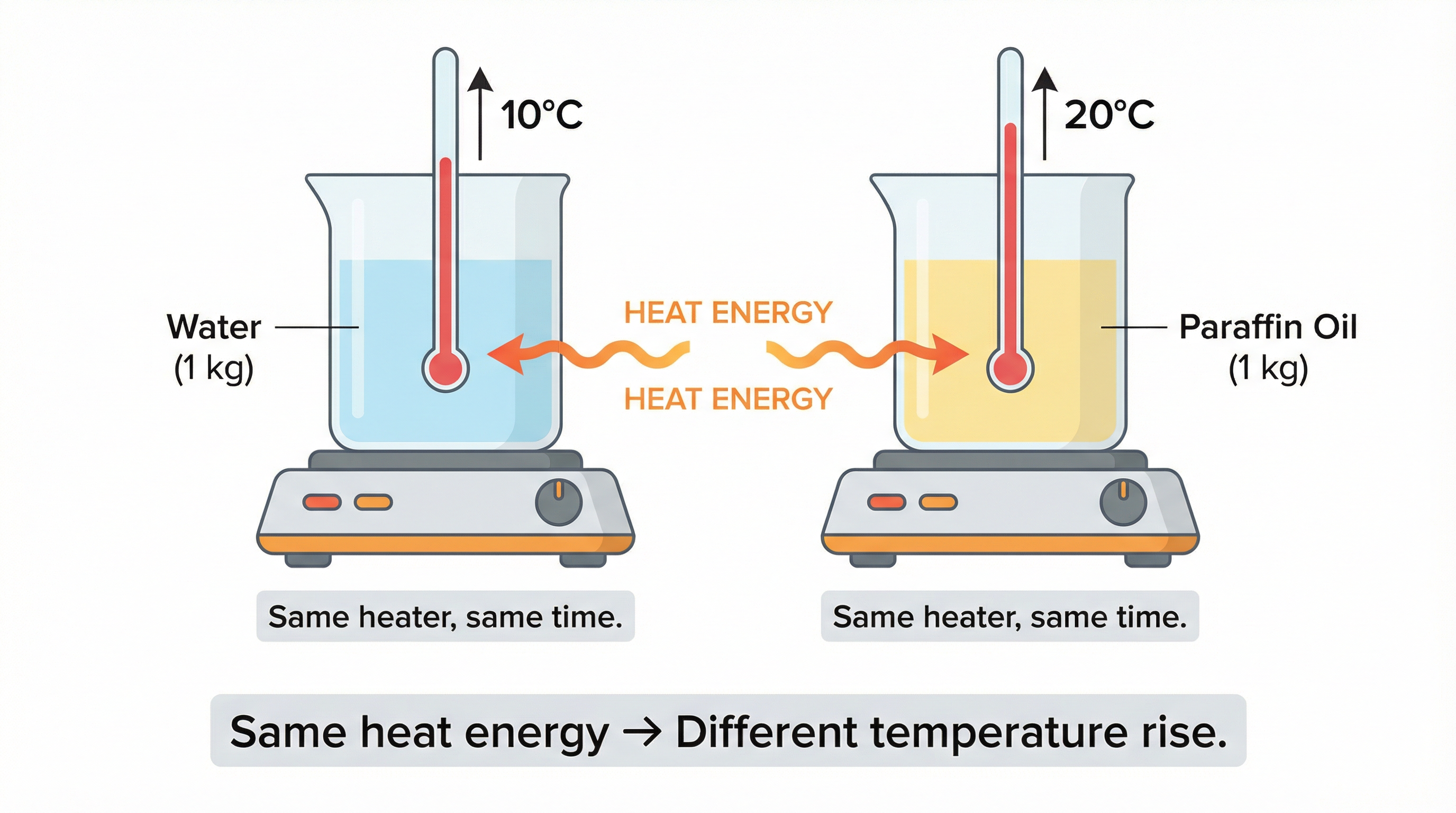
Fig.: Same heat energy supplied, but paraffin oil shows roughly double the temperature rise compared to water.
Definition: Specific Heat Capacity
Specific heat capacity is defined as the amount of heat per unit mass absorbed or given out by the substance to change its temperature by one unit (one degree) 1 °C or 1K.
Formula: Specific Heat Capacity
s = \[\frac {ΔQ}{(m · ΔT)}\]
Where,
SI Unit: J · kg⁻¹ · K⁻¹
CGS Unit: erg · g⁻¹ · °C⁻¹
Specific Heat Capacity — Data Table
The table below lists specific heat capacities at room temperature and atmospheric pressure. Notice how water dominates — about 10× higher than most metals.
| Substance | Category | s (J·kg⁻¹·K⁻¹) |
|---|---|---|
| Water | Liquid | 4186 |
| Ethanol | Liquid | 2500 |
| Paraffin oil | Liquid | 2130 |
| Kerosene | Liquid | 2118 |
| Aluminium | Metal | 903 |
| Glass | Solid | 837 |
| Carbon | Solid | 506.5 |
| Iron | Metal | 448 |
| Copper | Metal | 387 |
| Silver | Metal | 234 |
| Tungsten | Metal | 134.4 |
| Gold | Metal | 129 |
| Lead | Metal | 128 |
| Steel | Metal | 120 |
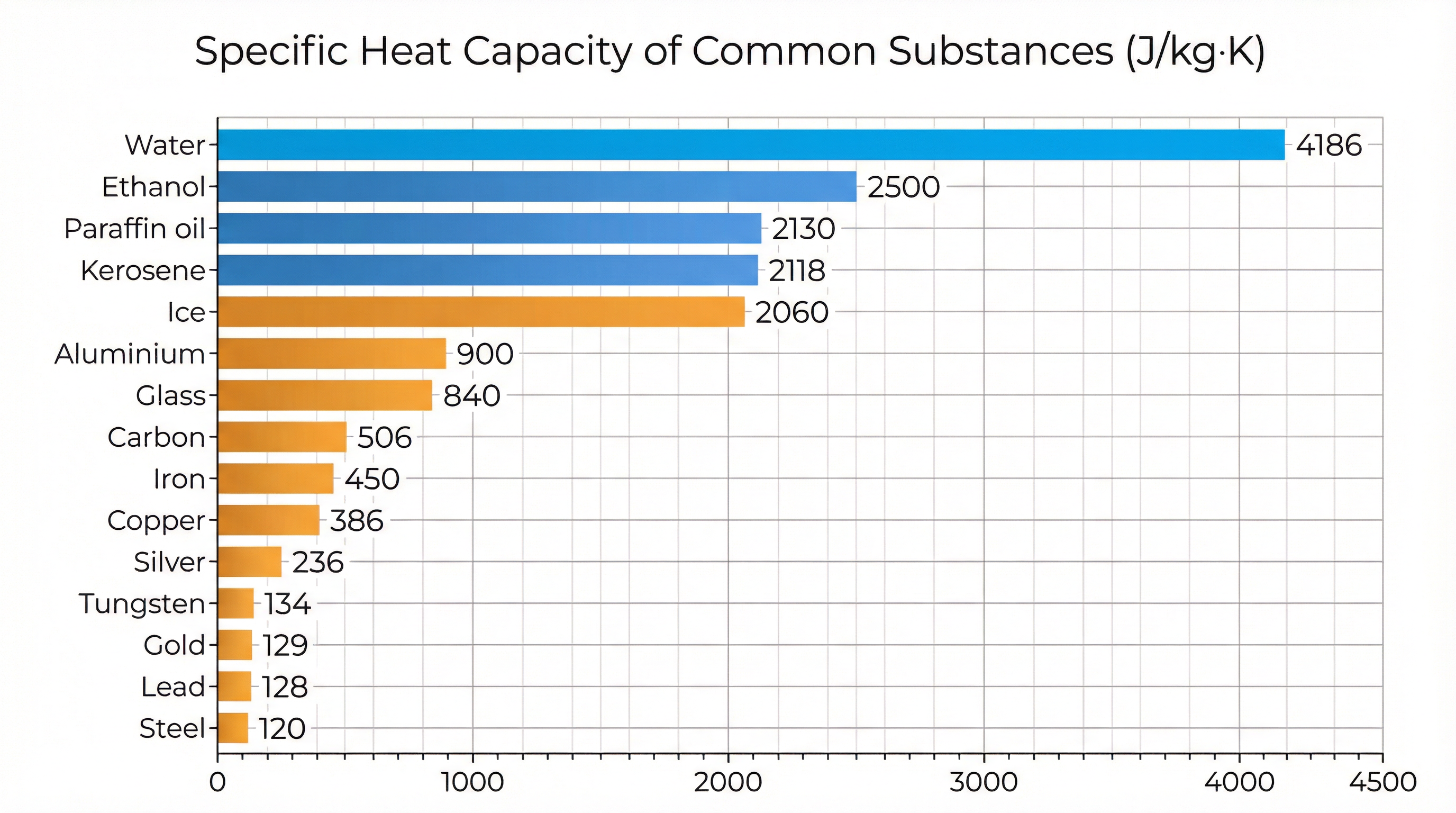
Fig.: Visual comparison: Water's specific heat capacity towers above all metals and most liquids.
Formula: Molar Specific Heat Capacity
When the amount of substance is measured in moles (μ) instead of kilograms, we use molar specific heat capacity (C):
C = \[\frac {1}{μ}\] · \[\frac {ΔQ}{ΔT}\]
SI Unit: J · mol⁻¹ · K⁻¹
Real-Life Applications
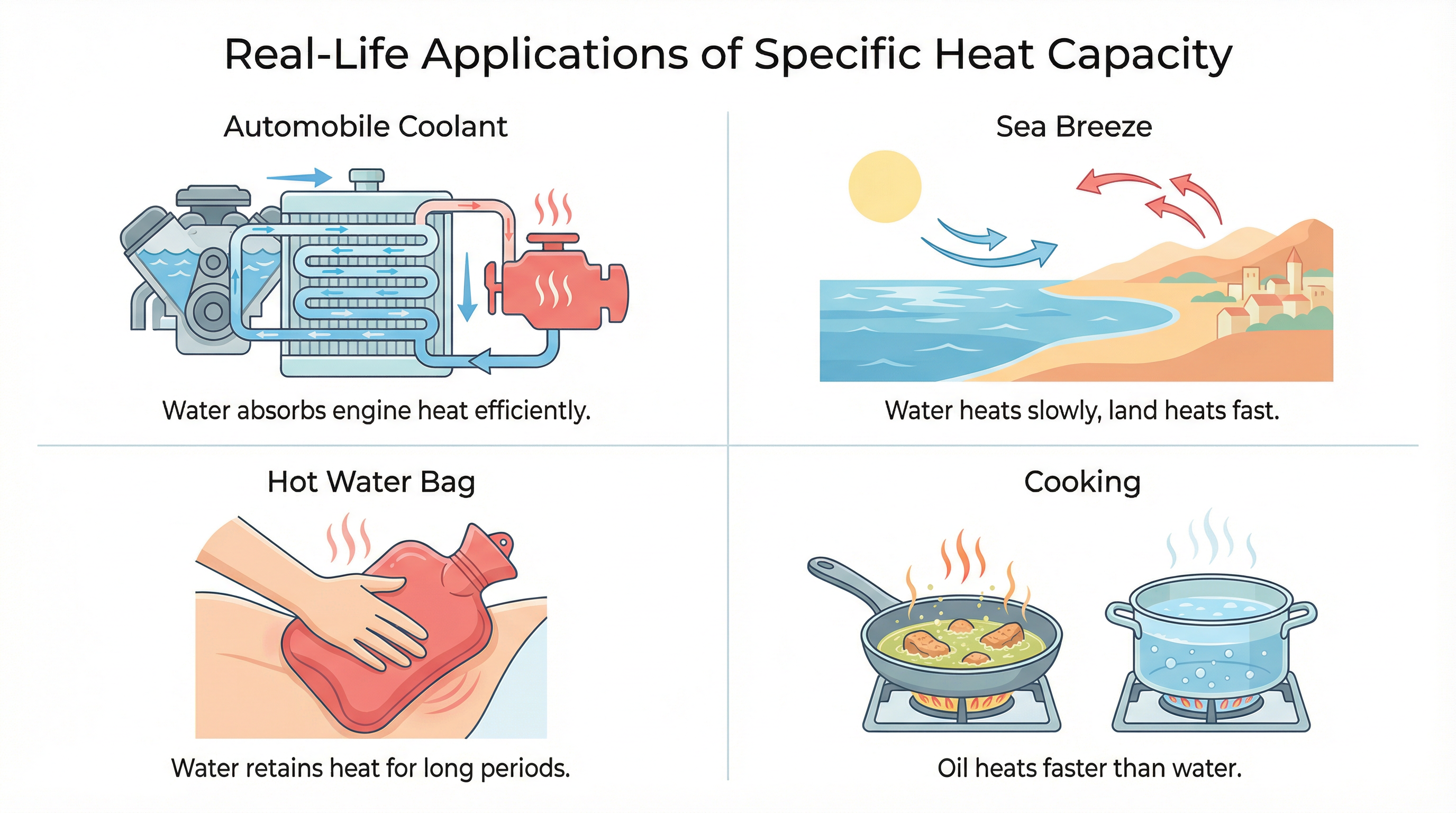 Fig.: Specific heat capacity at work in everyday life: from car engines to climate patterns.
Fig.: Specific heat capacity at work in everyday life: from car engines to climate patterns.
- Automobile Radiators: Water absorbs a lot of engine heat with only a small rise in temperature because it has a high specific heat.
- Hot Water Bags: Water stores heat for a long time, so hot water bags stay warm longer.
- Cooking (Water vs Oil): Oil heats food faster than water because it has a lower specific heat.
- Coastal Climate: Oceans heat and cool slowly, so coastal areas have moderate temperatures.
- Desert Climate: Sand heats and cools quickly, causing very hot days and cold nights.
Key Points: Specific Heat Capacity of Solids and Liquids
- Specific heat capacity (s) measures how much heat per unit mass is needed to change a substance's temperature by 1°C (or 1 K).
- Formula: ΔQ = m · s · ΔT — applies only when there is no phase change.
- Water has the highest specific heat (4186 J/kg·K) among common substances — this is why it's used in cooling systems and retains heat well.
- Molar specific heat (C) measures heat per mole. For gases, it splits into Cₚ (constant pressure) and Cᵥ (constant volume), with Cₚ > Cᵥ always.
- Specific heat is intrinsic; it depends on the material, not on the amount of substance taken.
- The concept explains everyday phenomena such as coastal climate moderation, engine cooling, cooking times, and therapeutic hot water bags.
