- Coastal Climate: Oceans heat slowly, keeping coastal temperatures mild and stable, while sand heats and cools quickly, causing extreme desert temperatures.
- Car Radiators: Water absorbs a lot of heat without overheating, making it ideal for cooling car engines.
- Cooking: Copper pans heat up quickly and spread heat evenly, while water in food takes longer to heat, slowing cooking.
Topics
Units and Measurements
- Quantitative Science
- System of Units
- Derived Quantities and Units
- Rules and Conventions for Writing SI Units and Their Symbols
- Measurement of Length
- Measurement of Mass
- Measurement of Time
- Dimensions and Dimensional Analysis
- Accuracy, Precision and Uncertainty in Measurement
- Errors in Measurements>Systematic Errors
- Errors in Measurements>Random Errors
- Estimation of Errors
- Combination of Errors
- Significant Figures
- Definitions of SI Units and Constants
Mathematical Methods
- Vector Analysis
- Scalar
- Vector
- Vector Operations>Multiplication of a Vector by a Scalar
- Vector Operations>Addition and Subtraction of Vectors
- Vector Operations>Triangle Law for Vector Addition
- Vector Operations>Law of parallelogram of vectors
- Resolution of Vectors
- Multiplication of Vectors
- Scalar Product(Dot Product)
- Vector Product (Cross Product)
- Concept of Calculus
- Differential Calculus
- Integral Calculus
Motion in a Plane
- Concept of Motion
- Rectilinear Motion
- Displacement
- Path Length
- Average Velocity
- Average Speed
- Instantaneous Velocity
- Instantaneous Speed
- Acceleration in Linear Motion
- Relative Velocity
- Motion in Two Dimensions - Motion in a Plane
- Average and Instantaneous Velocities
- Acceleration in a Plane
- Equations of Motion in a Plane with Constant Acceleration
- Relative Velocity in Two Dimensions
- Projectile Motion
- Uniform Circular Motion (UCM)
- Key Parameters of Circular Motion
- Centripetal Acceleration
- Conical Pendulum
Laws of Motion
- Fundamental Principles of Motion and Mechanics
- Types of Motion
- Aristotle’s Fallacy
- Newton’s Laws of Motion
- Newton's First Law of Motion
- Newton’s Second Law of Motion
- Newton's Third Law of Motion
- Inertial and Non-inertial Frames of Reference
- Types of Forces>Fundamental Forces in Nature
- Types of Forces>Contact and Non-Contact Forces
- Types of Forces>Real and Pseudo Forces
- Types of Forces>Conservative and Non-Conservative Forces
- Types of Forces>Work Done by a Variable Force
- Work Energy Theorem
- Principle of Conservation of Linear Momentum
- Collisions
- Elastic and Inelastic Collisions
- Perfectly Inelastic Collision
- Coefficient of Restitution e
- Expressions for Final Velocities in Elastic Head-On Collision
- Loss of Kinetic Energy in Perfectly Inelastic Head-On Collision
- Collision in Two Dimensions
- Impulse of a Force
- Necessity of Defining Impulse
- Rotational Analogue of a Force: Moment of a Force Or Torque
- Couple and Its Torque
- Proof of Independence of the Axis of Rotation
- Mechanical Equilibrium
- States of Equilibrium
- Centre of Mass>Mathematical Understanding of Centre of Mass
- Centre of Mass>Velocity of Centre of Mass
- Centre of Mass>Acceleration of Centre of Mass
- Centre of Mass>Characteristics of Centre of Mass
- Centre of Gravity
Gravitation
- Concept of Gravitation
- Kepler’s Laws
- Law of Orbit or Kepler's First Law
- Law of Areas or Kepler's Second Law
- Law of Periods or Kepler's Third Law
- Newton's Universal Law of Gravitation
- Measurement of the Gravitational Constant (G)
- Acceleration Due to Gravity (Earth’s Gravitational Acceleration)
- Variation in the Acceleration>Variation in Gravity with Altitude
- Variation in the Acceleration>Variation in Gravity with Depth
- Variation in the Acceleration>Variation in Gravity with Latitude and Rotation of the Earth
- Variation in the Acceleration>Effect of the shape of the Earth
- Gravitational Potential Energy
- Expression for Gravitational Potential Energy
- Connection of Potential Energy Formula with mgh
- Potential and Potential Difference
- Escape Velocity
- Earth Satellites
- Projection of Satellite
- Weightlessness in a Satellite
- Time Period of Satellite
- Binding Energy of an Orbiting Satellite
Mechanical Properties of Solids
- Mechanical Properties of Solids
- Elastic Behavior of Solids
- Stress and Strain
- Types of Stress and Corresponding Strain
- Hooke’s Law
- Elastic Modulus>Young’s Modulus
- Elastic Modulus>Bulk Modulus
- Elastic Modulus>Shear Modulus (Modulus of Rigidity)
- Elastic Modulus>Poisson’s Ratio
- Stress-strain Curve
- Strain Energy
- Hardness of Material
- Friction in Solids
- Origin of Friction
- Types of Friction>Static Friction
- Types of Friction>Kinetic Friction
- Types of Friction>Rolling Friction
Thermal Properties of Matter
- Thermal Properties of Matter
- Temperature and Heat
- Measurement of Temperature
- Absolute Zero and Absolute Temperature
- Ideal Gas Equation
- Thermal Expansion
- Linear Expansion
- Areal Expansion
- Volume Expansion
- Specific Heat Capacity of Solids and Liquids
- Relation Between Coefficient of Expansion
- Specific Heat Capacity of Gas
- Heat Equation
- Thermal Capacity
- Calorimetry
- Change of State
- Analysis of Observation>From Point A to B
- Analysis of Observation>From Point B to D
- Evaporation vs Boiling
- Boiling Point and Pressure
- Sublimation
- Phase Diagram
- Gas and Vapour
- Latent Heat
- Heat Transfer
- Conduction
- Thermal Conductivity
- Coefficient of Thermal Conductivity
- Thermal Resistance
- Applications of Thermal conductivity
- Convection
- Application of Convection
- Free and Forced Convection
- Radiation
- Newton’s Law of Cooling
Sound
- Sound Waves
- Common Properties of All Waves
- Transverse Waves
- Longitudinal Waves
- Mathematical Expression of a Wave
- The Speed of Travelling Waves
- The Speed of Transverse Waves
- The Speed of Longitudinal Waves
- Newton's Formula for Velocity of Sound
- Laplace’s Correction
- Factors Affecting Speed of Sound
- Principle of Superposition of Waves
- Echo
- Reverberation
- Acoustics
- Qualities of Sound
- Doppler Effect
- Source Moving and Listener Stationary
- Listener Approaching a Stationary Source with Velocity
- Both Source and Listener are Moving
- Common Properties between Doppler Effect of Sound and Light
- Major Differences between Doppler Effects of Sound and Light
Optics
- Fundamental Concepts of Light
- Nature of Light
- Ray Optics Or Geometrical Optics
- Cartesian Sign Convention
- Reflection>Reflection from a Plane Surface
- Reflection>Reflection from Curved Mirrors
- Total Internal Reflection
- Refraction of Light
- Applications of Total Internal Reflection
- Refraction at a Spherical Surface and Lenses
- Thin Lenses and Their Combination
- Refraction at a Single Spherical Surface
- Lens Makers' Equation
- Dispersion of Light
- Analysis of Prism
- Thin Prisms
- Some Natural Phenomena Due to Sunlight
- Defects of Lenses
- Optical Instruments
- Simple Microscope or a Reading Glass
- Compound Microscope
- Telescope
Electrostatics
- Concept of Electrostatics
- Electric Charge
- Basic Properties of Electric Charge
- Additive Nature of Charge
- Quantization of Charge
- Conservation of Charge
- Force between Charges
- Coulomb’s Law
- Scalar Form of Coulomb’s Law
- Relative Permittivity or Dielectric Constant
- Definition of Unit Charge from the Coulomb’s Law
- Coulomb's Law in Vector Form
- Principle of Superposition
- Electric Field
- Electric Field Intensity Due to a Point-Charge
- Practical Way of Calculating Electric Field
- Electric Lines of Force
- Electric Flux
- Gauss’s Law
- Electric Dipole
- Couple Acting on an Electric Dipole in a Uniform Electric Field
- Electric Intensity at a Point Due to an Electric Dipole
- Continuous Charge Distribution
Electric Current Through Conductors
- Concept of Electric Currents in Conductors
- Electric Current
- Flow of Current Through a Conductor
- Drift Speed
- Ohm's Law
- Limitations of Ohm’s Law
- Electrical Power
- Resistors
- Rheostat
- A combination of resistors in both series and parallel
- Specific Resistance
- Variation of Resistance with Temperature
- Electromotive Force (emf)
- Cells in Series
- Cells in Parallel
- Types of Cells
Magnetism
- Concept of Magnetism
- Magnetic Lines of Force
- The Bar Magnet
- Magnetic Field due to a Bar Magnet
- Magnetic Field Due to a Bar Magnet at an Arbitrary Point
- Gauss' Law of Magnetism
- The Earth’s Magnetism
Electromagnetic Waves and Communication System
- Foundations of Electromagnetic Theory
- EM Wave
- Sources of EM Waves
- Characteristics of EM Waves
- Electromagnetic Spectrum
- Radio Waves
- Microwaves
- Infrared waves
- Visible Light
- Ultraviolet rays
- X-rays
- Gamma Rays
- Propagation of EM Waves
- Ground (surface) Wave
- Space wave
- Sky wave propagation
- Communication System
- Elements of a Communication System
- Commonly Used Terms in Electronic Communication System
- Modulation
Semiconductors
- Concept of Semiconductors
- Electrical Conduction in Solids
- Band Theory of Solids
- Intrinsic Semiconductor
- Extrinsic Semiconductor
- n-type semiconductor
- p-type semiconductor
- Charge neutrality of extrinsic semiconductors
- p-n Junction
- Basics of Semiconductor Devices
- Applications of Semiconductors and P-n Junction Diode
- Thermistor
Estimated time: 10 minutes
- Formula: Heat Equation
- Example
- Real-World Applications
- Key Points: Heat Equation
Maharashtra State Board: Class 11
Formula: Heat Equation
The Heat Equation:
Q = m × c × ΔT
where ΔT = Tfinal − Tinitial
The quantity of heat absorbed or released by a substance depends on three factors:
- Mass (m): More mass requires more heat energy
- Temperature Change (ΔT): A bigger change needs more heat
- Nature of Substance (c): Different materials absorb heat differently
Maharashtra State Board: Class 11
Example
Problem: If the temperature of a 4 kg mass of a material with a specific heat capacity of 300 J/ kg °C rises from 20 °C to 30 °C. Find the heat received.
Solution:
Identify known values:
m = 4 kg, c = 300 J kg⁻¹ °C⁻¹, ΔT = 30 − 20 = 10°C
m = 4 kg, c = 300 J kg⁻¹ °C⁻¹, ΔT = 30 − 20 = 10°C
Apply the heat equation:
Q = m × c × ΔT
Q = m × c × ΔT
Q = 4 × 300 × 10
Q = 12,000 J = 12 kJ
Maharashtra State Board: Class 11
Real-World Applications
Specific heat capacity is not just a textbook formula — it explains everyday phenomena all around us.
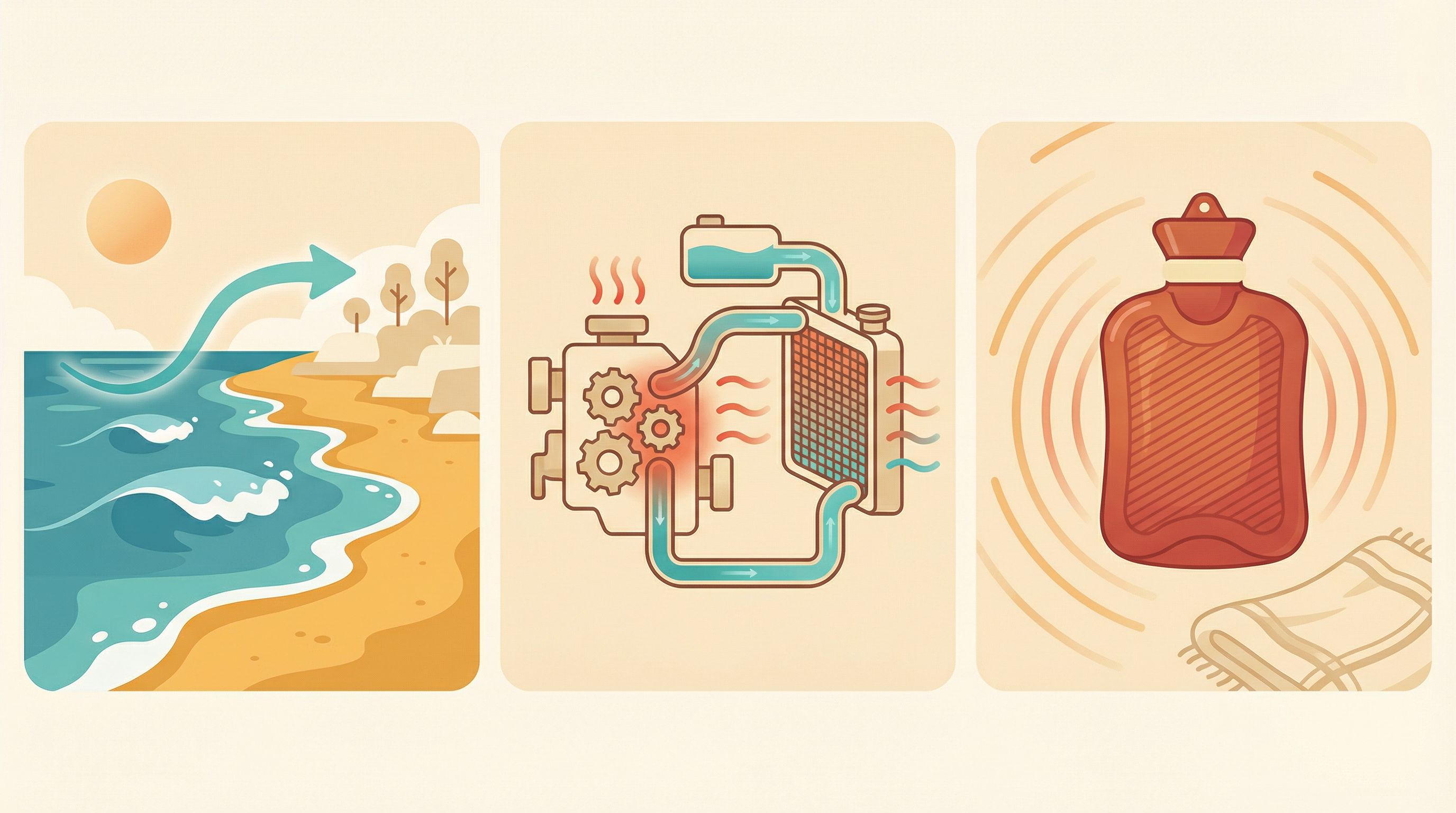
From ocean breezes to car engines — specific heat capacity shapes our daily experiences
Maharashtra State Board: Class 11
Key Points: Heat Equation
- Specific heat capacity tells us how "stubborn" a substance is to temperature changes — higher c means it's harder to heat or cool.
- The heat equation Q = m × c × ΔT is the fundamental formula connecting heat energy, mass, specific heat, and temperature change.
- Water has the highest specific heat (4186 J kg⁻¹ K⁻¹) among common substances — this regulates Earth's climate and is why water is used in cooling systems.
- The formula works in both directions: it calculates heat absorbed (temperature rise) or heat released (temperature fall).
- Units matter: always convert mass to kg and ensure ΔT is in °C or K (numerically identical for temperature changes).
