Definitions [50]
Biomolecules are organic compounds present in living organisms as essential constituents of different cells, such as carbohydrates, proteins, fats, and amino acids.
Define carbohydrates.
Carbohydrates are optically active polyhydroxy aldehydes or polyhydroxy ketones or compounds that can be hydrolysed to polyhydroxy aldehydes or polyhydroxy ketones.
Carbohydrates that are amorphous solids, tasteless and insoluble in water are catled non-sugars.
Carbohydrates that are crystalline solids, sweet in taste and soluble in water are called sugars.
Carbohydrates may be defined as optically active polyhydroxy aldehydes or ketones or compounds which produce such units on hydrolysis, such as cellulose, glycogen, starch, etc.
The sugars that reduce the Tollen's reagent and Fehling's solution are called reducing sugars.
A monosaccharide that contains one ketonic carbonyl group is called a ketose.
A ketose with six carbon atoms is called a ketohexose.
An aldose monosaccharide that has six carbon atoms (e.g., Glucose) is called an aldohexose.
Monosaccharides that contains one aldehydic group is called aldose.
Fructose is another commonly known monosaccharide having the same molecular formula as glucose. It is levorotatory and a ketohexose. It is present abundantly in fruits, and hence it is also called fruit sugar.
Define the term Protein.
Chemically proteins are polyamides which are high molecular weight polymers of the monomer units, i.e., α-amino acids. OR It can also be defined as proteins are the biopolymers of a large number of α-amino acids and they are naturally occurring polymeric nitrogenous organic compounds containing 16% nitrogen and peptide linkages (-CO-NH-)
Define α-amino acids.
α-Amino acids are carboxylic acids having an amino (–NH2) group bonded to the α-carbon, that is, the carbon next to the carboxyl (–COOH) group.
Define enzymes.
Enzymes are biological catalysts that speed up chemical reactions in living cells without being consumed in the process.
Define peptide bond.
The bond that connects α-amino acids to each other is called a peptide bond.
Proteins are complex polyamides formed from amino acids. They are essential for the proper growth and maintenance of the body. They have many peptide (-CO–NH )bonds.
Chemically, proteins are polyamides, which are high molecular weight polymers of the monomer units called \[\alpha\]-amino acids.
Bifunctional organic compounds containing a carboxylic and an amino group either at the same carbon atom or at nearby carbon atoms are called amino acids.
Define Zwitter ion
An ∝-amino acid molecule contains both acidic carboxyl (-COOH) group as well as basic amino (-NH2) group. Proton transfer from acidic group to basic group of amino acid forms a salt, which is a dipolar ion called zwitter ion.
Define the following term:
Essential amino acids
Amino acids that cannot be synthesised in the human body and must be obtained through diet are known as essential amino acids.
Amino acids which contain more number of carboxyl groups than amino groups are called acidic amino acids.
Amino acids which contain more number of amino groups than carboxyl groups are called basic amino acids.
Define the following term as related to proteins:
Primary structure
Proteins may have one or more polypeptide chains. Each polypeptide in a protein has amino acids linked with each other in a specific sequence and it is this sequence of amino acids that is said to be the primary structure of that protein. Any change in this primary structure, i.e., the sequence of amino acids, creates a different protein.
Amino acids which contain equal number of amino groups and carboxyl groups are called neutral amino acids.
Amino acids which are synthesised by the body itself are called non-essential amino acids.
Define the following as related to proteins:
Peptide linkage
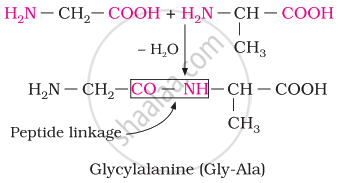
Chemically, peptide linkage is an amide formed between the –COOH group and –NH2 group. The reaction between two molecules of similar or different amino acids proceeds through the combination of the amino group of one molecule with the carboxyl group of the other. This results in the elimination of a water molecule and the formation of a peptide bond –CO–NH–. The product of the reaction is called a dipeptide because it is made up of two amino acids.
For example, when the carboxyl group of glycine combines with the amino group of alanine, we get a dipeptide, glycylalanine.
When a protein, in its native form, is exposed to changes, such as temperature or pH, the hydrogen bonds are disrupted. Due to this, globules unfold, the helix uncoils, and the protein loses its biological activity. It is called denaturation of proteins, e.g., coagulation of egg white on boiling, curdling of milk, etc.
Define the following term as related to proteins:
Denaturation
Denaturation is the process in which the secondary and tertiary structure of a protein is disrupted due to heat, a change in pH, or chemicals, while the primary structure remains unchanged. In denaturation, peptide bonds are not broken; only the weak bonds (like hydrogen bonds) are disturbed.
A colloidal solution of protein which works as a biological catalyst is known as an enzyme.
Vitamins are organic compounds essential for the average growth of life for animals, some bacteria and microorganisms.
Nucleic acids are large biological macromolecules that store and transmit genetic information in living organisms.
Define the following term.
nucleoside
The unit formed by joining the anomeric carbon of the furanose (sugar) with a nitrogen of a base is called nucleoside.
DNA is a double-stranded nucleic acid that stores and transmits hereditary information and can replicate itself.
A nucleotide is the basic structural unit of nucleic acids, composed of a nitrogenous base, a pentose sugar, and a phosphate group.
A nitrogenous base is an organic molecule (purine or pyrimidine) that carries genetic information in nucleic acids.
RNA is a single-stranded nucleic acid that helps in protein synthesis and information transfer.
A nucleoside consists of a nitrogenous base linked to a pentose sugar without a phosphate group.
The technique of identifying an individual by analyzing the unique DNA sequence present in each person, similar to fingerprints, is called DNA fingerprinting.
Biological catalysts which increase the rate of biochemical reactions are called enzymes.
Organic compounds required in small amounts in the diet to perform specific biological functions are called vitamins.
The unit formed when a nucleoside is linked to phosphoric acid is called nucleotide.
Long chains of nucleotides joined by phosphodiester linkage are called nucleic acids.
Chemical messengers secreted by endocrine glands and transported through blood are called hormones.
The sequence of amino acids in a polypeptide chain is called primary structure of protein.
The linkage formed between two monosaccharide units through an oxygen atom is called glycosidic linkage.
Carbohydrates which yield a large number of monosaccharide units on hydrolysis are called polysaccharides.
Carbohydrates which yield two to ten monosaccharide units on hydrolysis are called oligosaccharides.
Carbohydrates which cannot be hydrolysed to simpler units are called monosaccharides.
Optically active polyhydroxy aldehydes or ketones or the compounds which yield such units on hydrolysis are called carbohydrates.
The cyclic hemiacetal forms of a sugar differing in configuration at the anomeric carbon are called anomers.
Key Points
- Common Composition - All living organisms are made of the same elements, like carbon, hydrogen, and oxygen, detected through elemental analysis.
- Same Elements, Different Abundance - Both living and non-living matter contain the same elements, but carbon and hydrogen are more abundant in living organisms.
- Major Elements in Human Body - Oxygen (65%), Carbon (18.5%), Nitrogen (3.3%), Hydrogen (0.5%).
- Earth's Crust vs Human Body - Silicon (27.7%) and Oxygen (46.6%) dominate Earth's crust, while Carbon dominates living matter despite being only 0.03% in the crust.
- Significance - Living organisms selectively concentrate certain elements, making their composition different from non-living matter.
- Carbohydrates are organic biomolecules made of C, H and O, usually fitting the general formula Cx(H₂O)y and existing as aldoses or ketoses.
- They are classified into monosaccharides, disaccharides and polysaccharides; monosaccharides cannot be hydrolysed further, disaccharides are formed by two monosaccharides via glycosidic bonds, and polysaccharides are long polymers.
- Some sugars like digitoxose (C₆H₁₂O₄) and rhamnose (C₆H₁₂O₅) do not obey the typical Cx(H₂O)y formula.
- All monosaccharides are reducing sugars because they possess a free aldehyde or ketone group.
- Cellulose is a linear polymer of β‑D‑glucose, unlike starch and glycogen, which are polymers of α‑glucose and show branching.
- Biologically, carbohydrates supply energy for metabolism; glucose is the main substrate for ATP synthesis, and lactose provides energy to infants.
- Polysaccharides such as starch and glycogen act as storage products and also contribute to structural components of cell membranes and cell walls.
| Category | Meaning | Sub-classification | Examples |
|---|---|---|---|
| Monosaccharides | Simplest carbohydrates cannot be hydrolysed | Aldoses (contain –CHO group); Ketoses (contain C=O group) | Glucose (aldose), Fructose (ketose) |
| Disaccharides | Carbohydrates which on hydrolysis yield two monosaccharide units | Reducing sugars (have free –CHO group); Non-reducing sugars (no free –CHO group) | Maltose, Lactose (reducing); Sucrose (non-reducing) |
| Oligosaccharides | Carbohydrates which on hydrolysis yield 2–10 monosaccharide units | Includes trisaccharides, tetrasaccharides, etc. | Raffinose, Stachyose, Verbascose |
| Polysaccharides | Carbohydrates which on hydrolysis yield many monosaccharide units | Homopolysaccharides (same units); Heteropolysaccharides (different units) | Starch, Glycogen (homo); Cellulose, Chondroitin sulphate (hetero) |
- Glucose is a monosaccharide, an aldohexose, and a reducing sugar, commonly found in fruits and also known as dextrose.
- It can be prepared by hydrolysis of sucrose (using dilute acid) or hydrolysis of starch under heat and pressure.
- Glucose confirms a straight-chain structure of six carbon atoms when reduced to n-hexane.
- Presence of functional groups is shown by reactions: –CHO (aldehyde), five –OH groups, and formation of derivatives like oxime and cyanohydrin.
- Oxidation reactions indicate the formation of gluconic acid (mild oxidation) and saccharic acid (strong oxidation), confirming functional groups in glucose.
| Product | Inference |
|---|---|
| n-Hexane (hot HI) | 6 C in a straight chain |
| Glucoxime (NH₂OH) / Cyanohydrin (HCN) | Carbonyl group present |
| Gluconic acid (Br₂ water) | —CHO group present |
| Glucose pentaacetate (acetic anhydride) | Five —OH groups present |
| Saccharic acid (dil. HNO₃) | One primary —OH group present |
- Glucose is an aldohexose with molecular formula \[C_{6}H_{12}O_{6},\mathrm{M.P.146^{\circ}C.}\]
- 'D' in D-(+)-Glucose = configuration; (+) = dextrorotatory nature; 'D'/'L' have no relation to optical activity.
- Glucose has five —OH groups (confirmed by glucose pentaacetate) and one aldehydic carbonyl group (confirmed by oxime & cyanohydrin formation).
- Glucose is soluble in water, sparingly soluble in alcohol, and insoluble in ether.
- The additional chiral centre in glucose ring structures is formed due to ring closure.
Structure of Fructose:
| Open Chain Structure | Ring Structure | |
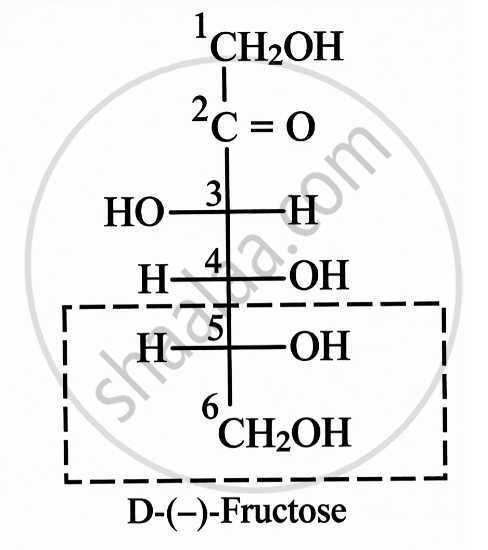 |
\[\alpha\]-D-(-)-Fructofuranose | \[\beta\]-D-(-)-Fructofuranose |
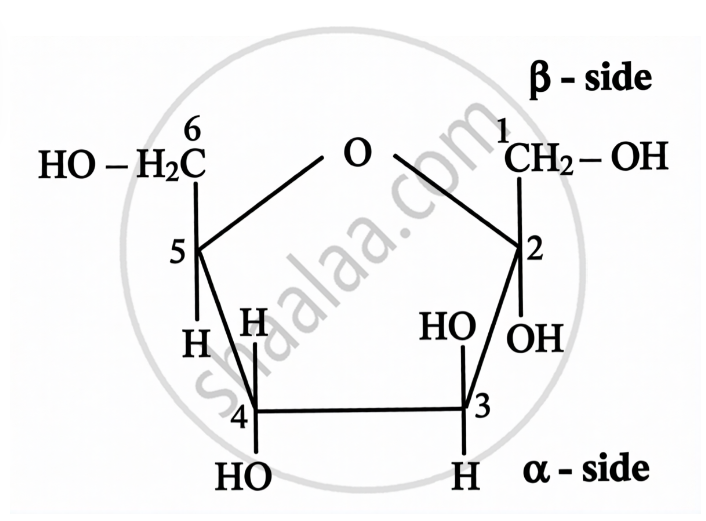 |
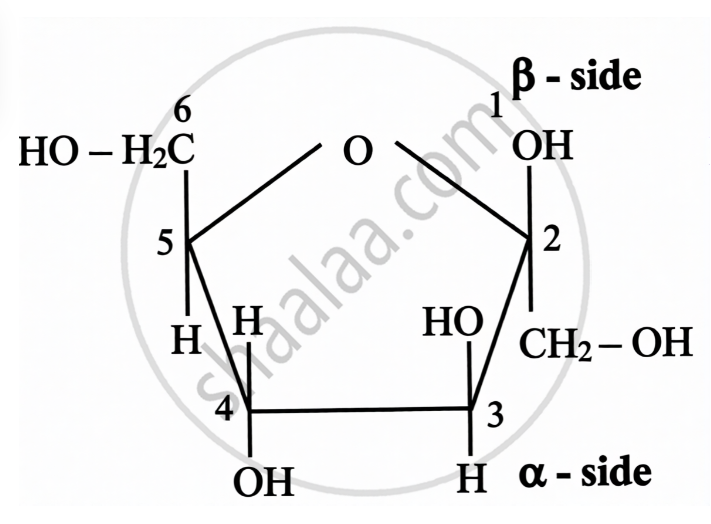 |
|
Preparation of Fructose:
-
From hydrolysis of cane sugar (sucrose):
\[ \underset{\text{Sucrose}}{\mathrm{C}_{12}\mathrm{H}_{22}\mathrm{O}_{11}} + \mathrm{H}_2\mathrm{O} \xrightarrow{\text{Dil } \mathrm{H}_2\mathrm{SO}_4} \underset{\text{Glucose}}{\mathrm{C}_6\mathrm{H}_{12}\mathrm{O}_6} + \underset{\text{Fructose}}{\mathrm{C}_6\mathrm{H}_{12}\mathrm{O}_6} \] -
From inulin:
\[ (\mathrm{C}_6\mathrm{H}_{10}\mathrm{O}_5)_n + n\mathrm{H}_2\mathrm{O} \xrightarrow{\text{Dil } \mathrm{H}_2\mathrm{SO}_4} \underset{\text{Fructose}}{n\mathrm{C}_6\mathrm{H}_{12}\mathrm{O}_6} \]
- Sucrose consists of one unit each of α-D-glucopyranose and β-D-fructofuranose.
- It contains an α, β-1,2-glycosidic linkage.
- Maltose is composed of two α-D-glucopyranose units joined by an α-1,4-glycosidic bond.
- Lactose consists of β-D-galactopyranose and β-D-glucopyranose units.
- It has a β-1,4-glycosidic linkage.
- Starch — A polymer of α-D-glucopyranose. It has two components: amylose (α-1,4-glycosidic linkage) and amylopectin (both α-1,4 and α-1,6-glycosidic linkages).
- Cellulose — A polymer of β-glucopyranose units linked by β-1,4-glycosidic bonds.
- Glycogen — A polymer of glucose units.
- Linkage Comparison —
Polysaccharide Monomer Linkage Amylose (Starch) α-D-glucopyranose α-1,4 Amylopectin (Starch) α-D-glucopyranose α-1,4 and α-1,6 Cellulose β-glucopyranose β-1,4 Glycogen Glucose — - Key Distinction — Starch and Cellulose are both glucose polymers but differ in linkage type: Starch has α-glycosidic bonds (digestible by humans), while Cellulose has β-glycosidic bonds (not digestible by humans).
- Primary source of energy for plants and animals
- Honey consists of a mixture of carbohydrates (source of quick energy)
- Starch and cellulose are stored in plants; glycogen is stored in animals/humans
- Used in the textile, paper, and alcohol industry
- Found in combination with proteins and lipids (e.g., glycoproteins, glycolipids)
- Proteins are polymers of amino acids (polypeptides) in which amino acids are linked by peptide bonds.
- There are 20 types of amino acids, so proteins are heteropolymers (not homopolymers).
- Amino acids are of two types: essential (must be obtained from diet) and non-essential (can be synthesised in the body).
- Proteins are high molecular weight biomolecules (polyamides) made of α-amino acids with a general structure R-CH(NH₂)-COOH.
- Proteins perform various functions such as enzymatic activity, transport, hormonal regulation, immunity, and sensory reception.
- Proteins are of two main types: fibrous proteins (insoluble, structural, e.g., keratin) and globular proteins (soluble, functional, e.g., enzymes, insulin).
- Collagen is the most abundant protein in animals, while RuBisCO is the most abundant enzyme in the biosphere.
α-Amino acids: Carboxylic acids where α-hydrogen is replaced by the –NH₂ group
General structure: \[ \mathrm{R} - \underset{\underset{\displaystyle \mathrm{NH}_2}{|}}{\overset{\overset{\displaystyle \alpha}{\displaystyle \mathrm{CH}}}{}} - \mathrm{COOH}\]
(where R = H or alkyl group at α-carbon)
Essential amino acids (not synthesised in the body; must be taken in food):
- Leucine
- Isoleucine
- Lysine
- Methionine
- Phenylalanine
- Threonine
- Tryptophan
- Valine
Non-essential amino acids (synthesised in the body):
- Alanine
- Asparagine
- Aspartic acid
- Cysteine
- Glutamic acid
- Glutamine
- Glycine
- Proline
- Serine
- Tyrosine
Semi-essential (50% body + 50% food):
- Arginine
- Histidine
Glycine is the only optically inactive amino acid (no chiral centre; R = H)
Isoelectric Point (pI):
- pH at which an amino acid does not migrate in an electric field → exists as a zwitterion (+NH₃–CH(R)–COO⁻)
- At pI, the concentration of zwitterions is maximum; anionic and cationic forms are equal
- At pI, an amino acid has the least solubility in water (used in separation by isoelectric precipitation)
- For neutral amino acid: \[pI=\frac{1}{2}(pk_{a_1}+pk_{a_2})\]
| Sr. No. | Name | R Group | 3-Letter | 1-Letter |
|---|---|---|---|---|
| 1 | Glycine | H– | Gly | G |
| 2 | Alanine | CH₃– | Ala | A |
| 3 | Valine* | Me₂CH– | Val | V |
| 4 | Leucine* | Me₂CH–CH₂– | Leu | L |
| 5 | Isoleucine* | CH₃–CH₂–CH(Me)– | Ile | I |
| 6 | Asparagine | H₂N–CO–CH₂– | Asn | N |
| 7 | Glutamine | H₂N–CO–CH₂–CH₂– | Gln | Q |
| 8 | Serine | HO–CH₂– | Ser | S |
| 9 | Threonine* | CH₃–CHOH– | Thr | T |
| 10 | Cysteine | HS–CH₂– | Cys | C |
| 11 | Methionine* | Me–S–CH₂–CH₂– | Met | M |
| 12 | Phenylalanine* | Ph–CH₂– | Phe | F |
| 13 | Tyrosine | p–HO–C₆H₄–CH₂– | Tyr | Y |
| 14 | Tryptophan* | Indole–CH₂– | Trp | W |
| 15 | Proline | Entire ring structure | Pro | P |
| 16 | Aspartic acid (Acidic) | HOOC–CH₂– | Asp | D |
| 17 | Glutamic acid (Acidic) | HOOC–CH₂–CH₂– | Glu | E |
| 18 | Lysine* (Basic) | H₂N–(CH₂)₄– | Lys | K |
| 19 | Arginine* (Basic) | HN=C(NH₂)–NH–(CH₂)₃– | Arg | R |
| 20 | Histidine* (Basic) | Imidazole–CH₂– | His | H |
| Level | Description | Stabilising Forces |
|---|---|---|
| Primary | Linear sequence of amino acids linked by peptide bonds | Peptide bonds |
| Secondary | α-helix (right-handed coil, –NH of one AA H-bonded to C=O of 4th residue) OR β-pleated sheet (when R group is small) | Hydrogen bonds |
| Tertiary | Further folding of secondary structure; 3D shape | Hydrophobic interactions, H-bonds, disulphide bonds, van der Waals, ionic interactions |
| Quaternary | Two or more polypeptide chains arranged spatially | All forces present in tertiary structure |
- Enzymes are biological catalysts, mostly proteins, that increase the rate of biochemical reactions without being consumed.
- Some enzymes are ribozymes, which are RNA molecules that act like enzymes.
- Enzymes have primary, secondary, and tertiary structures, and their 3D structure determines their specificity and function.
- Each enzyme has a specific active site where the substrate binds to form an enzyme–substrate complex.
- Enzymes are highly specific and lower the activation energy of reactions.
- Enzyme activity is affected by temperature and pH; most enzymes are denatured at high temperatures, while thermophilic enzymes remain stable at 80–90°C.
- Examples of enzymes include amylase (starch → glucose), pepsin (proteins → amino acids), lactase (lactose → glucose + galactose), and maltase (maltose → glucose).
Mechanism of Enzyme Action (Lock and Key model):
- Enzyme (E) binds to substrate (S) → ES complex (E + S → ES)
- Product formation: ES → EP
- Product released: EP → E + P (enzyme regenerated)
- Enzymes work best at 298 K to 313 K (25°C to 40°C) — optimum temperature
- Activity decreases with temperature increase or decrease beyond optimum range; stops at ~273 K
Vitamins = organic compounds essential in small amounts for normal growth and functioning
Not synthesised in adequate amounts by the body → must be supplied in the diet.
| Category | Vitamins | Key Feature |
|---|---|---|
| Fat-soluble | A (Retinol), D (Calciferol), E (Tocopherol), K (Phylloquinone) | Stored in the liver and adipose tissue; insoluble in water |
| Water-soluble | B complex (B₁, B₂, B₃, B₅, B₆, B₁₂), C | Transferred to blood frequently; not stored |
- Nucleic acids are biomacromolecules present in the acid-insoluble fraction and are responsible for the storage and transmission of genetic information (DNA and RNA).
- They are polynucleotides, formed by repeated units called nucleotides.
- Each nucleotide consists of three components: a nitrogenous base, a pentose sugar, and a phosphate group.
- Nitrogenous bases are of two types: purines (adenine, guanine) and pyrimidines (cytosine, thymine, uracil).
- The sugar present is either ribose (in RNA) or 2′-deoxyribose (in DNA).
- DNA is double-stranded and contains bases A, T, G, and C, while RNA is single-stranded and contains A, U, G, and C.
- DNA stores genetic information, while RNA plays a key role in protein synthesis and the expression of genetic information.
Nucleotide = Phosphate group + Sugar + Nitrogenous base
Hydrolysis pathway:
- DNA → Nucleotide → Phosphoric acid + Nucleoside → Sugar (2-deoxyribose) + Purine/Pyrimidine base
- RNA → Nucleotide → Phosphoric acid + Nucleoside → Sugar (Ribose) + Purine/Pyrimidine base
Nitrogen Bases:
| Type | DNA bases | RNA bases |
|---|---|---|
| Purines (double ring) | Adenine (A), Guanine (G) | Adenine (A), Guanine (G) |
| Pyrimidines (single ring) | Cytosine (C), Thymine (T) | Cytosine (C), Uracil (U) |
- Nitrogenous bases in nucleic acids are of two types: purines and pyrimidines.
- Purine bases have a double-ring structure and include Adenine (A) and Guanine (G).
- Pyrimidine bases have a single-ring structure and include Cytosine (C), Thymine (T), and Uracil (U).
- Thymine is present in DNA, while Uracil is present in RNA instead of thymine.
- DNA fingerprinting is a technique used to identify an individual by analysing the unique DNA pattern present in every person (except identical twins).
- It is based on satellite DNA, especially VNTRs (Variable Number Tandem Repeats) - short sequences repeated in tandem, whose number varies among individuals and creates DNA polymorphism.
- Principle: the differences in VNTR repeat number produce DNA fragments of different lengths, which appear as a unique banding pattern.
- Steps: DNA isolation → PCR amplification → restriction digestion → gel electrophoresis → Southern blotting → probe hybridisation → autoradiography → comparison of band patterns.
- Applications: forensic identification, paternity/maternity testing, pedigree studies, medical research, conservation biology, and evolutionary/anthropological studies.
Hormones are chemical substances produced by ductless (endocrine) glands; released into the bloodstream to regulate organ functions
Classified into: Steroid hormones, Amine hormones, Peptide hormones
| Type | Example | Gland | Function |
|---|---|---|---|
| Steroid | Testosterone | Testes | Development of male sex organs |
| Steroid | Oestrogen & Progesterone | Ovary | Female sex organ development; pregnancy |
| Steroid | Cortisone | Adrenal cortex | Regulates water and mineral salt metabolism |
| Amine | Adrenaline | Adrenal medulla | Increases blood pressure and pulse rate |
| Amine | Thyroxine | Thyroid | Stimulates oxidative metabolism; growth |
| Peptide | Oxytocin | Posterior pituitary | Contraction of uterus during childbirth |
| Peptide | Vasopressin | Posterior pituitary | Controls water reabsorption in kidneys |
| Peptide | Insulin | Pancreas (posterior pituitary) | Controls blood glucose (lowers) |
| Peptide | Glucagon | Pancreas | Increases blood glucose level |
| Vitamin | Main Sources | Deficiency Disease |
|---|---|---|
| Vitamin A | Fish liver oil, carrots, milk | Night blindness, xerophthalmia |
| Vitamin B₁ | Yeast, cereals, milk | Beri-beri |
| Vitamin B₂ | Milk, eggs | Cheilosis, skin disorders |
| Vitamin B₆ | Yeast, egg yolk, cereals | Convulsions |
| Vitamin B₁₂ | Meat, fish, eggs | Pernicious anaemia |
| Vitamin C | Citrus fruits, amla | Scurvy |
| Vitamin D | Sunlight, fish, egg yolk | Rickets, osteomalacia |
| Vitamin E | Vegetable oils | Weak muscles, fragile RBCs |
| Vitamin K | Green leafy vegetables | Delayed blood clotting |
- Proteins are classified into two types based on their molecular shape: fibrous proteins and globular proteins.
- Fibrous proteins consist of parallel polypeptide chains held together by hydrogen and disulphide bonds; they are generally insoluble in water and provide structural support.
- Globular proteins are formed when polypeptide chains coil into a spherical shape; they are usually soluble in water and perform functional roles such as enzymatic and hormonal activities.
- Protein structure is organized into four hierarchical levels: primary, secondary, tertiary, and quaternary, each representing increasing complexity of folding and organization.
- The stability of higher-level protein structures (secondary, tertiary, and quaternary) is maintained by hydrogen bonds, disulphide linkages, van der Waals forces, and electrostatic interactions.
Important Questions [53]
- What are the expected products of hydrolysis of lactose?
- The following questions are case-based questions. Read the passage carefully and answer the questions that follow:
- What are the hydrolysis products of sucrose?
- Which One of the Following is a Monosaccharide: Starch, Maltose, Fructose, Cellulose
- Write the Name of Two Monosaccharides Obtained on Hydrolysis of Lactose Sugar.
- Write the product obtained when D-glucose reacts with H2N − OH.
- Which Component of Starch is a Branched Polymer of α-glucose and Insoluble in Water?
- A 10% solution (by mass) of sucrose in water has freezing point of 269.15 K. Calculate the freezing point of 10% glucose in water, if freezing point of pure water is 273.15 K.
- Which of the following statements is not true about glucose?
- Glucose on Reaction with Hi Gives N-hexane. What Does It Suggest About the Structure of Glucose?
- Give the reaction of glucose with hydrogen cyanide. Presence of which group is confirmed by this reaction?
- When D-glucose reacts with HI, it forms ______.
- Answer the Following Question. What is the Basic Structural Difference Between Glucose and Fructose?
- Write the Product When D-glucose Reacts with Conc. Hno3.
- Differentiable Between the Following: Amylose and Amylopectin
- Write the reaction involved when D-glucose is treated with the following reagent: (CH3CO)2O
- Give a reason for the following observations: Penta-acetate of glucose does not react with hydroxylamine.
- Write the Reactions Involved When D-glucose is Treated with the Following Reagent: H2n-oh
- Write the reaction involved when D-glucose is treated with the following reagent: Br2 water
- Hydrolysis of sucrose is called ______.
- Answer the Following Question. Write the Products Obtained After the Hydrolysis of Lactose.
- What do you understand by the term glycosidic linkage?
- What is the Difference Between Acidic Amino Acids and Basic Amino Acids?
- Define the following term: Essential amino acids
- Amino acids show amphoteric behaviour. Why?
- Differentiable Between the Following: Essential Amino Acids Non-essential Amino Acids
- Give a reason for the following observations: Amino acids behave like salts.
- Draw the zwitter ion structure for sulphanilic acid.
- Define the following term as related to proteins: Primary structure
- Assertion (A): Proteins are polymers of α-amino acids connected by a peptide bond. Reason (R): A tetrapeptide contains 4 amino acids linked by 4 peptide bonds.
- Β-pleated sheet structure in proteins refers to ______.
- Write One Difference Between α-helix and β-pleated Structures of Proteins.
- Differentiate Between the Following : Peptide Linkage and Glycosidic Linkage
- Differentiate between the following: Fibrous proteins and Globular proteins
- An α-helix is a structural feature of ______.
- Proteins are polymers of ______.
- Write one example of a thermosetting polymer.
- Define the following term as related to proteins: Denaturation
- Define the Following Term: Denaturation of Protein
- Define the Following with an Example Denatured Protein
- Write one example of a Natural polymer.
- Why vitamin C cannot be stored in our body?
- Deficiency of Which Vitamin Causes Scurvy?
- Define the Following Term: Vitamins
- Give a reason for the following observations: Water soluble vitamins must be taken regularly in the diet.
- Assertion (A): Vitamin C cannot be stored in our body. Reason (R): Vitamin C is fat soluble and is excreted from the body in urine.
- Write the Product Formed When Glucose is Treated with Hi ?
- What Type of Linkage is Responsible for the Formation of Proteins?
- What products would be formed when a nucleotide from DNA containing thymine is hydrolysed?
- What is the Difference Between a Nucleoside and Nucleotide ?
- The Two Strands in DNA Are Not Identical but Are Complementary. Explain.
Concepts [26]
- Concept of Biomolecules
- Biomolecules in the Cell > Carbohydrates
- Classification of Carbohydrates
- Monosaccahrides
- Preparation of Glucose
- Structures of Glucose
- Fructose
- Disaccharides
- Polysaccharides
- Importance of Carbohydrates
- Biomolecules in the Cell > Proteins
- Amino Acids
- Classification of Amino Acids
- Structure of Proteins
- Denaturation of Proteins
- Biomolecules in the Cell > Enzymes
- Biomolecules in the Cell > Vitamins
- Classification of Vitamins
- Biomolecules in the Cell > Nucleic Acids
- Chemical Composition of Nucleic Acids
- Structure of Nucleic Acids
- Har Gobind Khorana
- DNA Fingerprinting
- Biological Functions of Nucleic Acids
- Biomolecules in the Cell > Hormones
- Overview of Biomolecules
