Advertisements
Advertisements
Question
Give the reaction of glucose with hydrogen cyanide. Presence of which group is confirmed by this reaction?
Advertisements
Solution
The reaction of glucose with hydrogen cyanide:
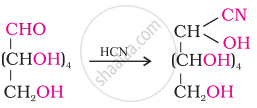
This reaction confirms the presence of the carbonyl group (>C = O) in glucose.
APPEARS IN
RELATED QUESTIONS
How many moles of acetic anhydride will be required to form glucose pentaacetate from 2M of glucose?
(a) 2
(b) 5
(c) 10
(d) 2.5
Answer the following question.
What is the basic structural difference between glucose and fructose?
Write the reactions involved when D-glucose is treated with the following reagent:
H2N-OH
The spatial arrangement of the given molecule is denoted by:

Acetylation of glucose yields ____________.
The symbols D and L represents ____________.
Glucose is found to exist in two different α and β crystalline forms. These forms can be obtained by:
(i) The α form of glucose is obtained by crystallisation from a concentrated solution of glucose at 303 K.
(ii) The β form of glucose is obtained by crystallisation from a concentrated solution of glucose at 303 K.
(iii) The β form is obtained by crystallisation from hot and saturated aqueous solution at 371 K.
(iv) The α form is obtained by crystallisation from hot and saturated aqueous solution at 371 K.
The reaction of glucose with red P + HI is called ____________.
The two forms of D-glucopyranose obtained from the solution of D-glucose are called ____________.
Which one of the following reactions is not explained by the open chain Structure of glucose?
