Advertisements
Advertisements
प्रश्न
Give the reaction of glucose with hydrogen cyanide. Presence of which group is confirmed by this reaction?
Advertisements
उत्तर
The reaction of glucose with hydrogen cyanide:
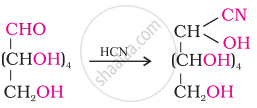
This reaction confirms the presence of the carbonyl group (>C = O) in glucose.
APPEARS IN
संबंधित प्रश्न
What happens when glucose is treated with hydroxylamine?
Differentiable between the following:
Amylose and Amylopectin
Oxime is formed by treating glucose with ____________.
Which of the following properties of glucose cannot be explained by its open chain structure?
(i) Glucose does not form hydrogen sulphite with NaHSO3.
(ii) On oxidation with HNO3 glucose gives saccharic acid.
(iii) Glucose is found to exist in two different crystalline forms which are named as α and β.
Reduction of glucose by HI suggest that ____________.
Which of the following pairs represents anomers?
Three structures are given below in which two glucose units are linked. Which of these linkages between glucose units are between C1 and C4 and which linkages are between C1 and C6?
| (I) |  |
| (II) |  |
| (III) |  |
How will you distinguish 1° and 2° hydroxyl groups present in glucose? Explain with reactions.
Assertion: D (+) – Glucose is dextrorotatory in nature.
Reason: ‘D’ represents its dextrorotatory nature.
The number of asymmetric carbon atoms in the glucose molecule in open and cyclic form is ______.
