Advertisements
Advertisements
प्रश्न
Enlist the properties of glucose that can not be explained on the basis of open chain structure of it
Advertisements
उत्तर
Although the open chain structure of D (+) − Glucose explains most of its reactions, it fails to explain the following facts:
a) D (+)-Glucose does not undergo certain characteristic reactions of aldehydes
eg., Glucose does not form NaHSO3 addition product, aldehyde-ammonia adduct 2, 4 − DNP derivative and does not respond to Schiff’s reagent test.
b) Glucose reacts with NH2OH to form an oxime but glucose pentaacetate does not react
with NH2OH, which implies that the free aldehyde group is absent in glucose pentaacetate
c) D(+)-Glucose exists in two stereoisomeric crystalline forms, i.e. α-glucose and
β-glucose, called anomers. α-D(+)-Glucose is obtained when a concentrated aqueous or
alcoholic solution is crystallised at 303 K. It has a melting point of 419 K and has a
specific rotation of +111° in a freshly prepared aqueous solution.
However, when glucose is crystallised from water above 371 K, β-D(+)-glucose is
obtained. It has a melting point of 423 K and has a specific rotation of +19.2° in a
freshly prepared aqueous solution. This behaviour could not be explained by the open
chain structure of glucose.
APPEARS IN
संबंधित प्रश्न
Enumerate the reactions of D-glucose which cannot be explained by its open chain structure.
Maltose is a
(a) Polysaccharide
(b) Disaccharide
(c) Trisaccharide
(d) Monosaccharide
Write the product when D-glucose reacts with conc. HNO3.
Glucose on reaction with HI gives n-hexane. What does it suggest about the structure of glucose?
What do you observe when glucose solution is heated with Tollen’s reagent?
Write the reactions involved when D-glucose is treated with the following reagent:
H2N-OH
Choose the appropriate answer(s) for the below representation from the options given

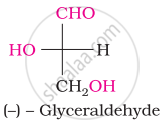
Glucose does not give Schiff’s test because of the formation of cyclic ____________.
The symbols D and L represents ____________.
Which one of the following compounds is different from the rest?
Glucose reacts with acetic anhydride to form ______.
Which of the following properties of glucose cannot be explained by its open chain structure?
(i) Glucose does not form hydrogen sulphite with NaHSO3.
(ii) On oxidation with HNO3 glucose gives saccharic acid.
(iii) Glucose is found to exist in two different crystalline forms which are named as α and β.
Reduction of glucose by HI suggest that ____________.
Which of the following reactions of glucose can be explained only by its cyclic structure?
Which is the least stable form of glucose?
A solution of D-glucose in water rotates the plane polarised light ____________.
The number of chiral carbon atoms present in cyclic structure α-D(+) glucose:
The two forms of D-glucopyranose obtained from the solution of D-glucose are called ____________.
The number of chiral carbons in ß-D(+) glucose is ____________.
Which one of the following reactions is not explained by the open chain Structure of glucose?
Why does compound (A) given below not form an oxime?

(A)
How will you distinguish 1° and 2° hydroxyl groups present in glucose? Explain with reactions.
Account for the following:
What happens when D – glucose is treated with the following reagents
Bromine water
Consider the following reactions:
(i) \[\ce{Glucose + R-OH ->[Conc. HNO3] [A] ->[X eq of][(CH3CO)2O] Acetyl derivative}\]
(ii) \[\ce{Glucose ->[Ni/H2] [A] ->[Y eq of][(CH3CO)2O] Acetyl derivative}\]
(iii) \[\ce{Glucose ->[Z eq of][(CH3CO)2O] Acetyl derivative}\]
'X, 'Y' and 'Z' in these reactions are respectively:
The number of asymmetric carbon atoms in the glucose molecule in open and cyclic form is ______.
When D-glucose reacts with HI, it forms ______.
Give the reaction of glucose with hydrogen cyanide. Presence of which group is confirmed by this reaction?
