Advertisements
Advertisements
प्रश्न
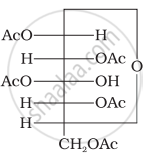
Why does compound (A) given below not form an oxime?

(A)
Advertisements
उत्तर
Glucose pentaacetate (structure A) does not have a free –OH group at C, and therefore, cannot be converted to the open chain form to give a free –CHO group and hence it does not form the oxime.
(Structure A)
APPEARS IN
संबंधित प्रश्न
Maltose is a
(a) Polysaccharide
(b) Disaccharide
(c) Trisaccharide
(d) Monosaccharide
Write the product when D-glucose reacts with conc. HNO3.
Fill in the blanks by choosing the appropriate word/words from those given in the brackets:
(iodoform, acetaldehyde, positive, greater, acidic, acetone, disaccharide, negative, increases, glucose, decreases, chloroform, polysaccharide, lactose, lesser, basic, cationic hydrolysis, anionic hydrolysis)
Sucrose is a _________ and yields upon hydrolysis, a mixture of ________ and fructose.
Answer the following question.
What is the basic structural difference between glucose and fructose?
Acetylation of glucose yields ____________.
Glucose does not give Schiff’s test because of the formation of cyclic ____________.
Which of the following statements is incorrect regarding glucose?
Glucose does not react with ____________.
Which of the following reactions of glucose can be explained only by its cyclic structure?
The α-D glucose and β-D glucose differ from each other due to difference in carbon atom with respect to its ____________.
Choose the correct relationship for glucose and fructose:
The number of chiral carbons in ß-D(+) glucose is ____________.
Which of the following pairs represents anomers?
Assertion: D (+) – Glucose is dextrorotatory in nature.
Reason: ‘D’ represents its dextrorotatory nature.
On the basis of which evidences D-glucose was assigned the following structure?
\[\begin{array}{cc}
\ce{CHO}\\
|\phantom{....}\\
\phantom{..}\ce{(CHOH)4}\\
|\phantom{....}\\
\phantom{..}\ce{CH2OH}
\end{array}\]
Account for the following:
What happens when D – glucose is treated with the following reagents
HNO3
Consider the following reactions:
(i) \[\ce{Glucose + R-OH ->[Conc. HNO3] [A] ->[X eq of][(CH3CO)2O] Acetyl derivative}\]
(ii) \[\ce{Glucose ->[Ni/H2] [A] ->[Y eq of][(CH3CO)2O] Acetyl derivative}\]
(iii) \[\ce{Glucose ->[Z eq of][(CH3CO)2O] Acetyl derivative}\]
'X, 'Y' and 'Z' in these reactions are respectively:
Glucose with excess of phenyl hydrazine forms ______.
The number of asymmetric carbon atoms in the glucose molecule in open and cyclic form is ______.
