Advertisements
Advertisements
प्रश्न
Account for the following:
What happens when D – glucose is treated with the following reagents
HNO3
Advertisements
उत्तर
\[\begin{array}{cc}
\phantom{......}\ce{CHO}\phantom{.............}\ce{COOH}\phantom{.............}\ce{COOH}\phantom{......}\\
\phantom{...}|\phantom{..................}|\phantom{...................}|\phantom{........}\\
\phantom{.}\ce{(CHOH)4 ->[Oxidation] (CHOH)4 <-[Oxidation] (CHOH)4}\\
\phantom{...}|\phantom{..................}|\phantom{...................}|\phantom{........}\\
\phantom{.....}\ce{CH2OH}\phantom{...........}\ce{\underset{\underset{acid}{Saccharic}}{COOH}}\phantom{.............}\ce{\underset{\underset{acid}{Gluconic}}{CH2OH}}\phantom{....}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Enlist the properties of glucose that can not be explained on the basis of open chain structure of it
What do you observe when glucose solution is heated with Tollen’s reagent?
Answer the following question.
What is the basic structural difference between glucose and fructose?
The number of asymmetric carbon atom(s) below the figure is/are

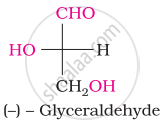
The reaction of glucose with red P + HI is called ____________.
The letter D and L in carbohydrates represent ____________.
Three structures are given below in which two glucose units are linked. Which of these linkages between glucose units are between C1 and C4 and which linkages are between C1 and C6?
| (I) |  |
| (II) |  |
| (III) |  |
Write the reactions of D-glucose which can’t be explained by its open-chain structure. How can cyclic structure of glucose explain these reactions?
Account for the following:
There are 5 OH groups in glucose
Give the reaction of glucose with hydrogen cyanide. Presence of which group is confirmed by this reaction?
