Advertisements
Advertisements
प्रश्न
Answer the following question.
What is the basic structural difference between glucose and fructose?
Advertisements
उत्तर
Both Glucose and Fructose are hexose sugars with six carbon atoms but Glucose is an aldohexose and fructose is ketohexose which means the functional group present in glucose is an aldehyde and the functional group in fructose is a ketone.
\[\begin{array}{cc}
\phantom{..}\ce{O}\phantom{..}\ce{H}\phantom{.}\ce{OH}\phantom{.}\ce{OH}\phantom{}\\
\phantom{}||\phantom{...}|\phantom{...}|\phantom{...}|\phantom{.}\\
\ce{CH2OH-C-C-C-C-CH2OH}\\
\phantom{....}|\phantom{...}|\phantom{...}|\phantom{}\\
\phantom{....}\ce{HO}\phantom{..}\ce{H}\phantom{..}\ce{H}\phantom{..}\\
\text{D-fructose}
\end{array}\]
\[\begin{array}{cc}
\phantom{}\ce{OH}\phantom{..}\ce{H}\phantom{.}\ce{OH}\phantom{.}\ce{OH}\phantom{....}\\
\phantom{.}|\phantom{...}|\phantom{...}|\phantom{...}|\phantom{....}\\
\ce{CHO-C-C-C-C-CH2OH}\\
\phantom{}|\phantom{...}|\phantom{...}|\phantom{...}|\phantom{...}\\
\phantom{}\ce{H}\phantom{.}\ce{HO}\phantom{.}\ce{H}\phantom{..}\ce{H}\phantom{..}\\
\text{D-glucose}
\end{array}\]
संबंधित प्रश्न
Choose the appropriate answer(s) for the below representation from the options given

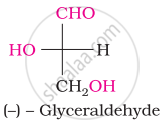
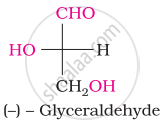
The number of asymmetric carbon atom(s) below the figure is/are

The number of chiral carbon atoms present in cyclic structure α-D(+) glucose:
Which one is correct?
Assertion: D (+) – Glucose is dextrorotatory in nature.
Reason: ‘D’ represents its dextrorotatory nature.
What happens when D-glucose is treated with the following reagent?
HI
Account for the following:
What happens when D – glucose is treated with the following reagents
Bromine water
Glucose with excess of phenyl hydrazine forms ______.
The number of asymmetric carbon atoms in the glucose molecule in open and cyclic form is ______.
Match List - I with List - II.
| List I | List II | ||
| (A) | Glucose + HI | (I) | Gluconic acid |
| (B) | Glucose + Br2 water | (II) | Glucose pentacetate |
| (C) | Glucose + acetic anhydride | (III) | Saccharic acid |
| (D) | Glucose + HNO3 | (IV) | Hexane |
Choose the correct answer from the options given below:
