Advertisements
Advertisements
प्रश्न
Answer the following question.
What is the basic structural difference between glucose and fructose?
Advertisements
उत्तर
Both Glucose and Fructose are hexose sugars with six carbon atoms but Glucose is an aldohexose and fructose is ketohexose which means the functional group present in glucose is an aldehyde and the functional group in fructose is a ketone.
\[\begin{array}{cc}
\phantom{..}\ce{O}\phantom{..}\ce{H}\phantom{.}\ce{OH}\phantom{.}\ce{OH}\phantom{}\\
\phantom{}||\phantom{...}|\phantom{...}|\phantom{...}|\phantom{.}\\
\ce{CH2OH-C-C-C-C-CH2OH}\\
\phantom{....}|\phantom{...}|\phantom{...}|\phantom{}\\
\phantom{....}\ce{HO}\phantom{..}\ce{H}\phantom{..}\ce{H}\phantom{..}\\
\text{D-fructose}
\end{array}\]
\[\begin{array}{cc}
\phantom{}\ce{OH}\phantom{..}\ce{H}\phantom{.}\ce{OH}\phantom{.}\ce{OH}\phantom{....}\\
\phantom{.}|\phantom{...}|\phantom{...}|\phantom{...}|\phantom{....}\\
\ce{CHO-C-C-C-C-CH2OH}\\
\phantom{}|\phantom{...}|\phantom{...}|\phantom{...}|\phantom{...}\\
\phantom{}\ce{H}\phantom{.}\ce{HO}\phantom{.}\ce{H}\phantom{..}\ce{H}\phantom{..}\\
\text{D-glucose}
\end{array}\]
संबंधित प्रश्न
Enumerate the reactions of D-glucose which cannot be explained by its open chain structure.
Write the product when D-glucose reacts with conc. HNO3.
What do you observe when glucose is treated with bromine water?
Write the reaction involved when D-glucose is treated with the following reagent:
(CH3CO)2O
The number of asymmetric carbon atom(s) below the figure is/are

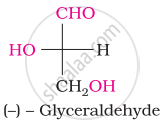
Which of the following statements is incorrect regarding glucose?
The letter D and L in carbohydrates represent ____________.
What happens when D-glucose is treated with the following reagent?
HI
Account for the following:
What happens when D – glucose is treated with the following reagents
Bromine water
The number of asymmetric carbon atoms in the glucose molecule in open and cyclic form is ______.
