Advertisements
Advertisements
प्रश्न
Account for the following:
What happens when D – glucose is treated with the following reagents
Bromine water
Advertisements
उत्तर
\[\begin{array}{cc}
\phantom{.}\ce{CHO}\phantom{..............}\ce{COOH}\phantom{..}\\
\phantom{.}|\phantom{..................}|\phantom{.......}\\
\phantom{}\ce{(CHOH)4 ->[Br2 water] (CHOH)4}\\
\phantom{.}|\phantom{..................}|\phantom{.......}\\
\phantom{...}\ce{CH2OH}\phantom{..........}\ce{\underset{Gluconic acid}{CH2OH}}\phantom{..}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
What do you observe when glucose solution is heated with Tollen’s reagent?
What do you observe when glucose is treated with bromine water?
Choose the appropriate answer(s) for the below representation from the options given

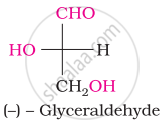
When glucose reacts with bromine water, the main product is ____________.
Which of the following reactions of glucose can be explained only by its cyclic structure?
Which is the least stable form of glucose?
The α-D glucose and β-D glucose differ from each other due to difference in carbon atom with respect to its ____________.
The two forms of D-glucopyranose obtained from the solution of D-glucose are called ____________.
The letter D and L in carbohydrates represent ____________.
Why does compound (A) given below not form an oxime?

(A)
