Advertisements
Advertisements
प्रश्न
Write the reaction involved when D-glucose is treated with the following reagent:
Br2 water
What happens when D-glucose is treated with the following reagent?
Bromine water
Advertisements
उत्तर
When glucose is heated with bromine water, it is oxidised to gluconic acid, a six-carbon carboxylic acid.
\[\begin{array}{cc}
\phantom{......}\ce{CHO}\phantom{.....................}\ce{COOH}\phantom{}\\
|\phantom{............................}|\phantom{..}\\
\phantom{.......}\ce{(CHOH)4->[Br2 water](CHOH)4}\\
|\phantom{............................}|\phantom{..}\\
\phantom{.............}\ce{\underset{\text{D-glucose}}{CH2OH}}\phantom{...............}\ce{\underset{\text{D-gluconic acid}}{CH2OH}}\phantom{....}
\end{array}\]
संबंधित प्रश्न
Write the reaction that indicates the presence of -CHO group in glucose
How many moles of acetic anhydride will be required to form glucose pentaacetate from 2M of glucose?
(a) 2
(b) 5
(c) 10
(d) 2.5
What do you observe when glucose solution is heated with Tollen’s reagent?
The spatial arrangement of the given molecule is denoted by:

Choose the appropriate answer(s) for the below representation from the options given

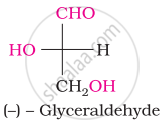
Acetylation of glucose yields ____________.
When glucose reacts with bromine water, the main product is ____________.
Glucose does not react with ____________.
Reduction of glucose by HI suggest that ____________.
A solution of D-glucose in water rotates the plane polarised light ____________.
The α-D glucose and β-D glucose differ from each other due to difference in carbon atom with respect to its ____________.
The two forms of D-glucopyranose obtained from the solution of D-glucose are called ____________.
The letter D and L in carbohydrates represent ____________.
Which of the following pairs represents anomers?
Why does compound (A) given below not form an oxime?

(A)
On the basis of which evidences D-glucose was assigned the following structure?
\[\begin{array}{cc}
\ce{CHO}\\
|\phantom{....}\\
\phantom{..}\ce{(CHOH)4}\\
|\phantom{....}\\
\phantom{..}\ce{CH2OH}
\end{array}\]
Account for the following:
There are 5 OH groups in glucose
Account for the following:
What happens when D – glucose is treated with the following reagents
Bromine water
Give a reason for the following observations:
Penta-acetate of glucose does not react with hydroxylamine.
