Advertisements
Advertisements
प्रश्न
Write the reaction that indicates the presence of -CHO group in glucose
Advertisements
उत्तर
The oxidation of glucose with bromine water (which is a mild oxidizing agent) forms gluconic acid. This indicates presence of aldehyde group.

APPEARS IN
संबंधित प्रश्न
Draw the simple Fisher projection formulae of D - (+) - glucose and D - (-) - fructose
How many moles of acetic anhydride will be required to form glucose pentaacetate from 2M of glucose?
(a) 2
(b) 5
(c) 10
(d) 2.5
Maltose is a
(a) Polysaccharide
(b) Disaccharide
(c) Trisaccharide
(d) Monosaccharide
Write the product when D-glucose reacts with conc. HNO3.
Write the reactions involved when D-glucose is treated with the following reagent:
H2N-OH
The following compound can be called as:

The spatial arrangement of the given molecule is denoted by:

Choose the appropriate answer(s) for the below representation from the options given

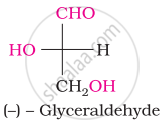
The number of asymmetric carbon atom(s) below the figure is/are

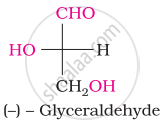
Oxime is formed by treating glucose with ____________.
Acetylation of glucose yields ____________.
The symbols D and L represents ____________.
Glucose does not react with ____________.
Glucose is found to exist in two different α and β crystalline forms. These forms can be obtained by:
(i) The α form of glucose is obtained by crystallisation from a concentrated solution of glucose at 303 K.
(ii) The β form of glucose is obtained by crystallisation from a concentrated solution of glucose at 303 K.
(iii) The β form is obtained by crystallisation from hot and saturated aqueous solution at 371 K.
(iv) The α form is obtained by crystallisation from hot and saturated aqueous solution at 371 K.
A solution of D-glucose in water rotates the plane polarised light ____________.
The number of chiral carbon atoms present in cyclic structure α-D(+) glucose:
Which one is correct?
Which one of the following reactions is not explained by the open chain Structure of glucose?
In the following reaction, identify A and B:
\[\begin{array}{cc}
\ce{C6H12O6 ->[Acetic anhydride] A}\\
\downarrow \text{Conc. nitric acid}\phantom{...}\\
\ce{B}\phantom{.................}\end{array}\]
Which of the following pairs represents anomers?
Write the reactions of D-glucose which can’t be explained by its open-chain structure. How can cyclic structure of glucose explain these reactions?
What happens when D-glucose is treated with the following reagent?
HI
Account for the following:
There are 5 OH groups in glucose
Account for the following:
What happens when D – glucose is treated with the following reagents
Bromine water
Account for the following:
What happens when D – glucose is treated with the following reagents
HNO3
The number of asymmetric carbon atoms in the glucose molecule in open and cyclic form is ______.
Give a reason for the following observations:
Penta-acetate of glucose does not react with hydroxylamine.
The reagents with which glucose does not react to give the corresponding tests/products are:
- Tollen’s reagent
- Schiff’s reagent
- HCN
- NH2OH
- NaHSO3
Choose the correct options from the given below:
