Advertisements
Advertisements
प्रश्न
On the basis of which evidences D-glucose was assigned the following structure?
\[\begin{array}{cc}
\ce{CHO}\\
|\phantom{....}\\
\phantom{..}\ce{(CHOH)4}\\
|\phantom{....}\\
\phantom{..}\ce{CH2OH}
\end{array}\]
Advertisements
उत्तर
This structure was assigned on the basis of the following evidences:
1. Molecular formula: The molecular formula of glucose has been found to be \[\ce{C6H12O6}\].
2. Straight chain structure:
(i) When aqueous solution of glucose is treated with sodium amalgam (Na/Hg) or sodium borohydride, it is reduced to sorbitol (or glucitol) a hexahedric alcohol.
\[\begin{array}{cc}
\phantom{.}\ce{CHO}\phantom{.......................}\ce{CH2OH}\phantom{..}\\
\phantom{}|\phantom{...........................}|\phantom{........}\\
\ce{(CHOH)4 + 2[H] ->[Na amalgam] (CHOH)4}\\
\phantom{}|\phantom{...........................}|\phantom{........}\\\
\phantom{..}\ce{CH2OH}\phantom{....................}\ce{\underset{Sorbitol}{CH2OH}\phantom{....}}
\end{array}\]
(ii) Prolonged heating with hydriodic acid and red phosphorus at 100°C gives a mixture of n-hexane and 2-iodohexane.
\[\begin{array}{cc}
\ce{\underset{Glucose}{CH2OH(CHOH)4CHO} ->[Hl][red P, 100°C] \underset{n-hexane}{CH3(CH2)4CH3} + CH3CH(CH2)3CH3}\\
\phantom{............................................}|\\
\phantom{............................................}\ce{\underset{2-Iodohexane}{I}}
\end{array}\]
The formation of n-hexane suggests that all the six carbon atoms in glucose are arranged in a straight chain structure of glucose.
3. Presence of five hydroxyl (-OH) groups: On acetylation with acetic anhydride, glucose gives a pentaacetate. This confirms that glucose contains five –OH groups. We know that the presence of two or more –OH groups on the same carbon atom makes the molecules unstable. Now since glucose is a stable compound, therefore, the five -OH groups must present on different carbon atoms.
4. Presence of one primary alcoholic group: On oxidation with cone, nitric acid, both glucose and gluconic acid give the same dicarboxylic acid, saccharic acid or glucaric acid. The primary alcoholic group \[\ce{(CH2OH)}\] is always present at the end of the carbon chain.
5. Presence of an aldehyde (-CHO) group: Glucose reacts with hydroxylamine, \[\ce{NH2OH}\] to form glucose CHO oxime. Which suggest that glucose contains a carbonyl \[\ce{(CHOH)4}\] (>C = O) groups.
APPEARS IN
संबंधित प्रश्न
Write the reaction that indicates the presence of -CHO group in glucose
Draw the simple Fisher projection formulae of D - (+) - glucose and D - (-) - fructose
Write the product when D-glucose reacts with conc. HNO3.
Differentiable between the following:
Amylose and Amylopectin
Answer the following question.
What is the basic structural difference between glucose and fructose?
Write the reaction involved when D-glucose is treated with the following reagent:
Br2 water
Choose the appropriate answer(s) for the below representation from the options given

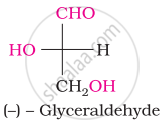
Glucose does not give Schiff’s test because of the formation of cyclic ____________.
Glucose is found to exist in two different α and β crystalline forms. These forms can be obtained by:
(i) The α form of glucose is obtained by crystallisation from a concentrated solution of glucose at 303 K.
(ii) The β form of glucose is obtained by crystallisation from a concentrated solution of glucose at 303 K.
(iii) The β form is obtained by crystallisation from hot and saturated aqueous solution at 371 K.
(iv) The α form is obtained by crystallisation from hot and saturated aqueous solution at 371 K.
The reaction of glucose with red P + HI is called ____________.
Which is the least stable form of glucose?
The letter D and L in carbohydrates represent ____________.
Which one is correct?
Which one of the following reactions is not explained by the open chain Structure of glucose?
Three structures are given below in which two glucose units are linked. Which of these linkages between glucose units are between C1 and C4 and which linkages are between C1 and C6?
| (I) |  |
| (II) |  |
| (III) |  |
Consider the following reactions:
(i) \[\ce{Glucose + R-OH ->[Conc. HNO3] [A] ->[X eq of][(CH3CO)2O] Acetyl derivative}\]
(ii) \[\ce{Glucose ->[Ni/H2] [A] ->[Y eq of][(CH3CO)2O] Acetyl derivative}\]
(iii) \[\ce{Glucose ->[Z eq of][(CH3CO)2O] Acetyl derivative}\]
'X, 'Y' and 'Z' in these reactions are respectively:
The number of asymmetric carbon atoms in the glucose molecule in open and cyclic form is ______.
When D-glucose reacts with HI, it forms ______.
