Advertisements
Advertisements
प्रश्न
Glucose on reaction with HI gives n-hexane. What does it suggest about the structure of glucose?
Advertisements
उत्तर
The reaction of glucose with HI giving n-hexane suggests that all the six carbon atoms are linked in a straight chain, as shown in the reaction given below:

APPEARS IN
संबंधित प्रश्न
Write the reaction that indicates the presence of -CHO group in glucose
Maltose is a
(a) Polysaccharide
(b) Disaccharide
(c) Trisaccharide
(d) Monosaccharide
Write the product when D-glucose reacts with conc. HNO3.
Choose the appropriate answer(s) for the below representation from the options given

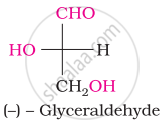
The number of asymmetric carbon atom(s) below the figure is/are

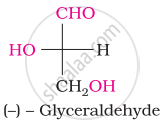
Which of the following statements is incorrect regarding glucose?
The α-D glucose and β-D glucose differ from each other due to difference in carbon atom with respect to its ____________.
Which one is correct?
In the following reaction, identify A and B:
\[\begin{array}{cc}
\ce{C6H12O6 ->[Acetic anhydride] A}\\
\downarrow \text{Conc. nitric acid}\phantom{...}\\
\ce{B}\phantom{.................}\end{array}\]
Glucose with excess of phenyl hydrazine forms ______.
