Advertisements
Advertisements
Question
Why does compound (A) given below not form an oxime?

(A)
Advertisements
Solution
Glucose pentaacetate (structure A) does not have a free –OH group at C, and therefore, cannot be converted to the open chain form to give a free –CHO group and hence it does not form the oxime.
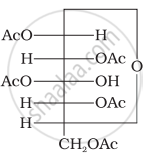
(Structure A)
APPEARS IN
RELATED QUESTIONS
What happens when glucose is treated with hydroxylamine?
Maltose is a
(a) Polysaccharide
(b) Disaccharide
(c) Trisaccharide
(d) Monosaccharide
Write the product when D-glucose reacts with conc. HNO3.
Glucose on reaction with HI gives n-hexane. What does it suggest about the structure of glucose?
Fill in the blanks by choosing the appropriate word/words from those given in the brackets:
(iodoform, acetaldehyde, positive, greater, acidic, acetone, disaccharide, negative, increases, glucose, decreases, chloroform, polysaccharide, lactose, lesser, basic, cationic hydrolysis, anionic hydrolysis)
Sucrose is a _________ and yields upon hydrolysis, a mixture of ________ and fructose.
Write the reactions involved when D-glucose is treated with the following reagent:
H2N-OH
Choose the appropriate answer(s) for the below representation from the options given

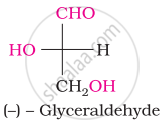
The number of asymmetric carbon atom(s) below the figure is/are

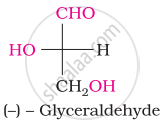
The symbols D and L represents ____________.
Glucose does not react with ____________.
Which is the least stable form of glucose?
A solution of D-glucose in water rotates the plane polarised light ____________.
Which one of the following reactions is not explained by the open chain Structure of glucose?
Three structures are given below in which two glucose units are linked. Which of these linkages between glucose units are between C1 and C4 and which linkages are between C1 and C6?
| (I) |  |
| (II) |  |
| (III) |  |
What happens when D-glucose is treated with the following reagent?
HI
Account for the following:
What happens when D – glucose is treated with the following reagents
Bromine water
The number of asymmetric carbon atoms in the glucose molecule in open and cyclic form is ______.
The reagents with which glucose does not react to give the corresponding tests/products are:
- Tollen’s reagent
- Schiff’s reagent
- HCN
- NH2OH
- NaHSO3
Choose the correct options from the given below:
