Advertisements
Advertisements
Question
Maltose is a
(a) Polysaccharide
(b) Disaccharide
(c) Trisaccharide
(d) Monosaccharide
Advertisements
Solution
Disaccharide
APPEARS IN
RELATED QUESTIONS
Enlist the properties of glucose that can not be explained on the basis of open chain structure of it
Glucose on reaction with HI gives n-hexane. What does it suggest about the structure of glucose?
Differentiable between the following:
Amylose and Amylopectin
Fill in the blanks by choosing the appropriate word/words from those given in the brackets:
(iodoform, acetaldehyde, positive, greater, acidic, acetone, disaccharide, negative, increases, glucose, decreases, chloroform, polysaccharide, lactose, lesser, basic, cationic hydrolysis, anionic hydrolysis)
Sucrose is a _________ and yields upon hydrolysis, a mixture of ________ and fructose.
What do you observe when glucose is treated with bromine water?
Answer the following question.
What is the basic structural difference between glucose and fructose?
Write the reactions involved when D-glucose is treated with the following reagent:
H2N-OH
Choose the appropriate answer(s) for the below representation from the options given

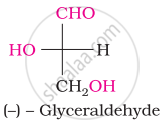
The symbols D and L represents ____________.
Which one of the following compounds is different from the rest?
Glucose does not react with ____________.
Glucose is found to exist in two different α and β crystalline forms. These forms can be obtained by:
(i) The α form of glucose is obtained by crystallisation from a concentrated solution of glucose at 303 K.
(ii) The β form of glucose is obtained by crystallisation from a concentrated solution of glucose at 303 K.
(iii) The β form is obtained by crystallisation from hot and saturated aqueous solution at 371 K.
(iv) The α form is obtained by crystallisation from hot and saturated aqueous solution at 371 K.
Reduction of glucose by HI suggest that ____________.
The reaction of glucose with red P + HI is called ____________.
Which of the following reactions of glucose can be explained only by its cyclic structure?
The number of chiral carbon atoms present in cyclic structure α-D(+) glucose:
Choose the correct relationship for glucose and fructose:
The letter D and L in carbohydrates represent ____________.
The number of chiral carbons in ß-D(+) glucose is ____________.
Which one of the following reactions is not explained by the open chain Structure of glucose?
Which of the following pairs represents anomers?
Why does compound (A) given below not form an oxime?

(A)
Assertion: D (+) – Glucose is dextrorotatory in nature.
Reason: ‘D’ represents its dextrorotatory nature.
Write the reactions of D-glucose which can’t be explained by its open-chain structure. How can cyclic structure of glucose explain these reactions?
Consider the following reactions:
(i) \[\ce{Glucose + R-OH ->[Conc. HNO3] [A] ->[X eq of][(CH3CO)2O] Acetyl derivative}\]
(ii) \[\ce{Glucose ->[Ni/H2] [A] ->[Y eq of][(CH3CO)2O] Acetyl derivative}\]
(iii) \[\ce{Glucose ->[Z eq of][(CH3CO)2O] Acetyl derivative}\]
'X, 'Y' and 'Z' in these reactions are respectively:
Glucose with excess of phenyl hydrazine forms ______.
Give a reason for the following observations:
Penta-acetate of glucose does not react with hydroxylamine.
