Advertisements
Advertisements
प्रश्न
Enumerate the reactions of D-glucose which cannot be explained by its open chain structure.
Advertisements
उत्तर १
- Aldehydes give 2, 4-DNP test, Schiff’s test, and react with NaHSO3 to form the hydrogen sulphite addition product. However, glucose does not undergo these reactions.
- The pentaacetate of glucose does not react with hydroxylamine. This indicates that a free −CHO group is absent from glucose.
- Glucose exists in two crystalline forms – α and β. The α-form (m.p. = 419 K) crystallises from a concentrated solution of glucose at 303 K and the β-form (m.p. = 423 K) crystallises from a hot and saturated aqueous solution at 371 K. This behaviour cannot be explained by the open chain structure of glucose.
उत्तर २
D-(+)-glucose does not undergo certain characteristic reactions of aldehydes, e.g., glucose does not form NaHSO3 addition product.
Glucose reacts with NH2OH to form an oxime but glucose pentaacetate does not. This implies that the aldehydic group is absent in glucose pentaacetate.
D-(+)-glucose exists in two stereoisomeric forms, i.e., α-glucose and β-glucose.
Both α-D-glucose and β-D-glucose undergo mutarotation in aqueous solution. Although the crystalline forms of α- and β-D-(+)-glucose are quite stable in aqueous solution, each form slowly changes into an equilibrium mixture of both.
D-(+)-glucose forms two isomeric methyl glucosides. Aldehydes normally react with two moles of methanol per mole of the aldehyde to form an acetal but D-(+)-glucose, when treated with methanol in the presence of dry HCl gas, reacts with only one mole of methanol per mole of glucose to form a mixture of two methyl D-glucosides i.e., methyl-α-D-glucoside (melting point 438 K, specific rotation +158°) and methyl-β-D-glucoside (melting point 308 K, specific rotation – 33°).
APPEARS IN
संबंधित प्रश्न
Draw the simple Fisher projection formulae of D - (+) - glucose and D - (-) - fructose
What happens when glucose is treated with hydroxylamine?
What do you observe when glucose solution is heated with Tollen’s reagent?
Write the reaction involved when D-glucose is treated with the following reagent:
Br2 water
The spatial arrangement of the given molecule is denoted by:

The number of asymmetric carbon atom(s) below the figure is/are

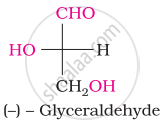
Which one of the following compounds is different from the rest?
The α-D glucose and β-D glucose differ from each other due to difference in carbon atom with respect to its ____________.
The two forms of D-glucopyranose obtained from the solution of D-glucose are called ____________.
The number of chiral carbons in ß-D(+) glucose is ____________.
In the following reaction, identify A and B:
\[\begin{array}{cc}
\ce{C6H12O6 ->[Acetic anhydride] A}\\
\downarrow \text{Conc. nitric acid}\phantom{...}\\
\ce{B}\phantom{.................}\end{array}\]
Assertion: D (+) – Glucose is dextrorotatory in nature.
Reason: ‘D’ represents its dextrorotatory nature.
What happens when D-glucose is treated with the following reagent?
HI
Account for the following:
What happens when D – glucose is treated with the following reagents
HNO3
Glucose with excess of phenyl hydrazine forms ______.
Match List - I with List - II.
| List I | List II | ||
| (A) | Glucose + HI | (I) | Gluconic acid |
| (B) | Glucose + Br2 water | (II) | Glucose pentacetate |
| (C) | Glucose + acetic anhydride | (III) | Saccharic acid |
| (D) | Glucose + HNO3 | (IV) | Hexane |
Choose the correct answer from the options given below:
Give the reaction of glucose with hydrogen cyanide. Presence of which group is confirmed by this reaction?
