Advertisements
Advertisements
प्रश्न
Give the reaction of glucose with hydrogen cyanide. Presence of which group is confirmed by this reaction?
Advertisements
उत्तर
The reaction of glucose with hydrogen cyanide:
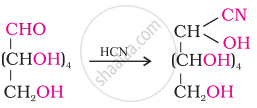
This reaction confirms the presence of the carbonyl group (>C = O) in glucose.
APPEARS IN
संबंधित प्रश्न
Draw the simple Fisher projection formulae of D - (+) - glucose and D - (-) - fructose
Enumerate the reactions of D-glucose which cannot be explained by its open chain structure.
Write the product when D-glucose reacts with conc. HNO3.
Glucose on reaction with HI gives n-hexane. What does it suggest about the structure of glucose?
The spatial arrangement of the given molecule is denoted by:

Oxime is formed by treating glucose with ____________.
Acetylation of glucose yields ____________.
What happens when D-glucose is treated with the following reagent?
HI
Account for the following:
What happens when D – glucose is treated with the following reagents
HNO3
The reagents with which glucose does not react to give the corresponding tests/products are:
- Tollen’s reagent
- Schiff’s reagent
- HCN
- NH2OH
- NaHSO3
Choose the correct options from the given below:
