Advertisements
Advertisements
Question
Explain D and L configuration in sugars.
Advertisements
Solution
D and L configuration in sugars:
- Conventionally (+)-glyceraldehyde is represented by the Fischer projection formula having –OH group attached to C-2 on the right side and this configuration is denoted by symbol ‘D’.
- Similarly, the configuration of (–) glyceraldehyde is denoted by the symbol ‘L’.
- All the compounds which can be correlated by a series of chemical reactions to (+)-glyceraldehyde are said to have D-configuration.
- And compounds which are chemically correlated to (–)-glyceraldehyde are said to have L-configuration. This is the system of the relative configuration of chiral compounds.
- A monosaccharide is assigned D/L configuration on the basis of the configuration of the lowest chiral carbon in its Fischer projection formula.
- Relative configuration of (+)-glucose with respect to (+)-glyceraldehyde can be drawn as follows:

APPEARS IN
RELATED QUESTIONS
Draw a neat diagram for the following:
Haworth formula of glucopyranose.
Glucose on oxidation with dilute nitric acid gives _______________
The glycosidic linkage in maltose is formed between _______________
Write the name of the unit to which glucose unit is linked to from sucrose.
Write chemical reaction for following conversions
glucose into gluconic acid
Classify the following carbohydrates into monosaccharide, disaccharide, oligosaccharide and polysaccharide:
glucose, cellulose, maltose, stachyose
What is the action of the following reagents on glucose?
hydroxylamine
What is the action of the following reagents on glucose?
hydrogen iodide
By which of the following process formation of glycosidic bond occurs?
Identify the bond that links the carbon of two adjacent monosaccharides.
From the following identify an example of disaccharides.
Which of the following monosaccharides is a tetrose sugar?
From the following which is a heteropolysaccharide?
______ is the prosthetic group of glycoproteins.
Identify the sugar having the molecular formula C6H1206.
Which among the following compounds is obtained when glucose reacts with hydrogen cyanide?
By which of the following feature we can identify the relatively small DNA molecules of plasmids?
Raffinose, sucrose and stachyose are respectively ____________.
Which one of the following is an oligosaccharide?
\[\ce{CH3 - CH2 - CH2 - Br ->[Na][dry ether] A ->[Cl2] B}\]
Identify the product B in the above reaction.
Which one of the following is generally applicable to polysaccharides?
Which one of the following carbohydrates is insoluble in water?
Which of the following molecules reduces Fehling's solution?
Which one of the following sugar does NOT have same empirical fonnula as that of carbohydrate?
Identify the number of secondary carbon atoms in glucose.
Which among the following reagents is used to obtain gluconic acid from glucose?
How many moles of acetic acid are obtained in the reaction when one mole glucose is treated with excess acetic anhydride?
What is the product obtained when Br2 water reacts with glucose?
When one mole of lactose is hydrolysed, the hydrolysate contains ____________.
The correct corresponding order of names of four aldoses with configuration given below Respectively is:
Complete hydrolysis of cellulose gives ____________.
α-D (+) Glucose and β-D (+) glucose are ____________.
What are reducing and non-reducing sugars?
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Fructose
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Sucrose
Is the following sugar, D-sugar or L-sugar?

Which of the following groups contain polysaccharides?
Lactose is made of ______.
Glucose on oxidation with bromine water yields gluconic acid. This reaction confirms the presence of ______.
Corn is immersed in boiling water. It is then cooled, and the solution becomes sweet. It is due to ______.
Which one is a non-reducing commercial sugar?
Identify the product obtained in the following conversion.
\[\ce{Glucose ->[(O)][Br2 water] Product}\]
Why carbohydrates are generally optically active.
Assign D/L configuration to the following monosaccharides:
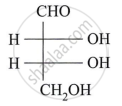
Carbohydrates that do not undergo hydrolysis further are called ______.
Write Fischer projection formulae for glucose.
Write the ring structure of glucose.
CH2 OH - CO - (CHOH)4 - CH2 OH is an example of ______.
The linkage present in Lactose is ______.
CH2 OH - CO - (CHOH)4 - CH2 OH is an example of ______.
Why are carbohydrates generally optically active?
Write the Zwitter ion structure of alanine.
Why carbohydrates are generally optically active?
Why carbohydrates are generally optically active?
