Advertisements
Advertisements
Question
Write chemical reaction for following conversions
glucose into gluconic acid
Advertisements
Solution
Glucose gets oxidised to a six carbon monocarboxylic acid called gluconic acid on reaction with bromine water which is a mild oxidizing agent.
\[\begin{array}{cc}
\phantom{}\ce{CHO}\phantom{...................}\ce{COOH}\phantom{.}\\
\phantom{}|\phantom{.......................}|\phantom{......}\\
\phantom{}\ce{(CHOH)4 + [O] ->[Br2 water]\phantom{}(CHOH)4}\phantom{}\\
\phantom{}|\phantom{.......................}|\phantom{......}\\
\phantom{..}\ce{\underset{\text{Glucose}}{CH2OH} \phantom{...............}\underset{\text{Gluconic acid}}{CH2OH}}\phantom{}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Give scientific reasons:
The disaccharide sucrose gives negative Tollens test while the disaccharide maltose gives a positive Tollens test.
Write chemical reaction for following conversions
glucose into glucoxime
Explain the classification of carbohydrates with examples.
Define carbohydrates.
Which of the following type of carbohydrates cannot be hydrolyzed?
Identify the given structure 'P' and 'Q'.

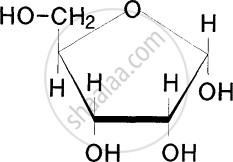
Identify the substances having glycosidic bond and peptide bond, respectively in their structure:
Which of the following monosaccharides is a tetrose sugar?
Formation of lactose by conden ation results in the release of how many molecules of water?
The reserve food material in animals is ____________.
Identify the sugar having the molecular formula C6H1206.
From the following identify the materials that are made up of cellulose.
i. Plant cell wall
ii. Exoskeleton of arthropods
iii. Paper from plant pulp
iv. Cotton fibre
Monosaccharides are ______ in nature.
Which reagent among the following is used to confirm presence of aldehydic carbonyl group in glucose?
Which carbon atoms of α- D glucopyranose and β-D-fructofuranose respectively are linked together to form glycosidic linkage in sucrose?
All these carbohydrates contain \[\ce{1 -> 4β}\] glycosidic linkage, EXCEPT ____________.
One mole of a carbohydrate on hydrolysis gives one mole of glucose and one mole of galactose. Identify the carbohydrate.
Which element among the following is not present in saccharine?
Which one of the following is an oligosaccharide?
\[\ce{CH3 - CH2 - CH2 - Br ->[Na][dry ether] A ->[Cl2] B}\]
Identify the product B in the above reaction.
Which among the following statements is true for amylose?
When 2 moles of stachyose is hydrolyzed, the number of moles of galactose obtained is ____________.
Which among the following reagents is used to confirm the presence of carbonyl group in glucose?
Which of the following statement is NOT true about saccharic acid?
On hydrolysis sucrose gives ____________.
Identify the number of oxygen atoms present in saccharic acid?
Identify the product Y in the following reaction.
\[\ce{Maltose ->[Hydrolysis] X ->[dil. HNO3][(excess)] Y}\]
The number of sp2 and sp3 hybridised carbon in fructose are respectively ____________.
Glucose is an aldose. Which one of the following reactions is not expected with glucose?
Which of the following are epimers?
Why carbohydrates are generally optically active?
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Starch
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Maltose
A molecule of stachyose contains how many carbon atoms?
The two monosaccharides in a disaccharide are held together by ______ bonds.
Match the Column I with Column II and choose the correct answer from options below:
| Column I | Column II |
| A. Purine | 1. Glycogen |
| B. Pyrimidine | 2. Cellulose |
| C. Structural polysaccharide | 3. Glucagon |
| D. Storage polysaccharide | 4. Adenine |
| 5. Cytosine |
Identify the monosaccharide containing only one asymmetric carbon atom in its molecule.
Which one is a non-reducing commercial sugar?
Formation of gluconic acid from glucose by oxidation using Br2 water.
Consider the following reaction
\[\ce{A <-[Br2 - H2O] Glucose ->[HNO3] B}\]
Here, 'A' and 'B' are respectively.
\[\ce{CH2OH - CO - (CHOH)4 - CH2OH}\] is an example of ______.
Why carbohydrates are generally optically active.
Describe the action of the following reagent on glucose:
Bromine water
Carbohydrates that do not undergo hydrolysis further are called ______.
Explain the hydrolysis of sucrose.
CH2 OH - CO - (CHOH)4 - CH2 OH is an example of ______.
Why carbohydrates are generally optically active.
Why carbohydrates are generally optically active.
The linkage present in Lactose is ______.
Why carbohydrates are generally optically active.
\[\ce{CH2OH-CO-(CHOH)4-CH2OH}\] is an example of ______.
Why carbohydrates are generally optically active.
Given below are two statements.
Statement I - Lactose on hydrolysis converts into glucose and galactose.
Statement II - Galactose also plays same role as glucose in respiration.
In light of the above statements, select the correct option given below.
