Advertisements
Advertisements
Question
What are reducing and non-reducing sugars?
Advertisements
Solution
1. Reducing sugars: Those carbohydrates which contain free aldehyde or ketonic group and reduces Fehling’s solution and Tollen’s reagent are called reducing sugars. All monosaccharides whether aldose or ketone are reducing sugars.
2. Non-reducing sugars: Carbohydrates that do not reduce Tollen’s reagent and Fehling’s solution are called non – reducing sugars. Example Sucrose. They do not have a free aldehyde group.
APPEARS IN
RELATED QUESTIONS
\[\ce{CH2OH-CO-(CHOH)4-CH2OH}\] is an example of ______.
Give scientific reasons:
The disaccharide sucrose gives negative Tollens test while the disaccharide maltose gives a positive Tollens test.
Draw a neat diagram for the following:
Haworth formula of maltose
The glycosidic linkage in maltose is formed between _______________
Write the name of the unit to which glucose unit is linked to from sucrose.
What is the action of the following reagents on glucose?
acetic anhydride
What is the action of the following reagents on glucose?
hydroxylamine
Define carbohydrates.
Write a commercial method for preparation of glucose.
Draw the structure of the pyran.
Which of the following type of carbohydrates cannot be hydrolyzed?
Identify the bond that links the carbon of two adjacent monosaccharides.
Identify the given structure 'P' and 'Q'.

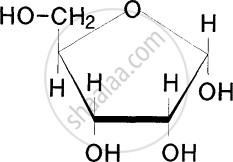
Identify the substances having glycosidic bond and peptide bond, respectively in their structure:
Which enzymes would work sequentially on potatoes consumed by an individual?
Carbohydrates can contain which of the following chemical groups?
From the following identify the group that is exclusively consists of polysaccharides.
From the following which is a heteropolysaccharide?
The reserve food material in animals is ____________.
From the following identify the materials that are made up of cellulose.
i. Plant cell wall
ii. Exoskeleton of arthropods
iii. Paper from plant pulp
iv. Cotton fibre
Identify the WRONG statement.
By which of the following feature we can identify the relatively small DNA molecules of plasmids?
Identify a non-reducing carbohydrate from the following.
Which is the product obtained, when Br2 water reacts with glucose?
From the following identify the two types of glucose polymers present in starch.
4-O-(α-D-Glucopyranosyl)-D-glucopyranose is ____________.
Which element among the following is not present in saccharine?
Which one of the following is generally applicable to polysaccharides?
Which one of the following is Tetrose sugar?
Which one of the following is NOT soluble in water?
Which among the following statements is true for amylose?
What is the quantity of glucose obtained when 68.4 g of sucrose is hydrolyzed in laboratory under ideal condition?
(Molar mass of sucrose = 342 g mol−1)
Which among the following reagents is used to obtain gluconic acid from glucose?
Which among the following is a product of hydrolysis of one mole raffinose?
What is the product obtained when Br2 water reacts with glucose?
How many hydroxyl groups are present in Erythrulose?
Which among the following observations suggests that glucose also exists in cyclic form?
Which one of the following rotates the plane polarized light towards left?
The correct corresponding order of names of four aldoses with configuration given below Respectively is:
Which one given below is a non-reducing sugar?
\[\ce{Glucose ->[(HCN)] Product ->[(hydrolysis)] Product ->[(HI + Heat)] A}\], the compound A is:
The number of sp2 and sp3 hybridised carbon in fructose are respectively ____________.
Glucose is an aldose. Which one of the following reactions is not expected with glucose?
α-D (+) Glucose and β-D (+) glucose are ____________.
Why carbohydrates are generally optically active?
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Starch
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Sucrose
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Lactose
The molecule of glucose is also called ______.
A molecule of stachyose contains how many carbon atoms?
The two monosaccharides in a disaccharide are held together by ______ bonds.
Corn is immersed in boiling water. It is then cooled, and the solution becomes sweet. It is due to ______.
Starch and cellulose are compounds made up of many units of ______.
\[\ce{CH2OH-CO-(CHOH)4-CH2OH}\] is an example of ______.
Assign D/L configuration to the following monosaccharides:
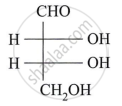
Describe the action of the following reagent on glucose:
dil. Nitric acid.
Write Fischer projection formulae for glucose.
The linkage present in Lactose is ______.
Why are carbohydrates generally optically active?
Why carbohydrates are generally optically active.
Why carbohydrates are generally optically active.
Given below are two statements.
Statement I - Lactose on hydrolysis converts into glucose and galactose.
Statement II - Galactose also plays same role as glucose in respiration.
In light of the above statements, select the correct option given below.
