Advertisements
Advertisements
Question
What is the action of the following reagents on glucose?
hydroxylamine
Advertisements
Solution
Glucose forms oxime by reaction with hydroxylamine:
\[\begin{array}{cc}
\phantom{}\ce{CHO}\phantom{..............}\ce{CH = N - OH}\phantom{..}\\
\phantom{.}|\phantom{..................}|\phantom{...............}\\
\ce{(CHOH)4 ->[NH2 - OH](CHOH)4 + H2O}\\
\phantom{}|\phantom{..................}|\phantom{..............}\\
\phantom{}\ce{\underset{\text{Glucose}}{CH2OH} \phantom{..........}\underset{\text{Oxime}}{CH2OH}}\phantom{........}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Write the name of the polysaccharide used for the commercial preparation of glucose.
Explain the classification of carbohydrates with examples.
Define carbohydrates.
Draw the Haworth projection structure of the following.
α-D-(–)-Fructofuranose
Draw the structure of the pyran.
Which of the following type of carbohydrates cannot be hydrolyzed?
Which enzymes would work sequentially on potatoes consumed by an individual?
Which of the following is the basic unit of carbohydrates?
From the following identify the group that is exclusively consists of polysaccharides.
______ is the prosthetic group of glycoproteins.
Identify the WRONG statement.
Which among the following compounds is obtained when glucose reacts with hydrogen cyanide?
Identify a non-reducing carbohydrate from the following.
Chitin is a/an ______.
From the following identify the two types of glucose polymers present in starch.
Identify the CORRECT combination.
Which carbon atoms of α- D glucopyranose and β-D-fructofuranose respectively are linked together to form glycosidic linkage in sucrose?
All these carbohydrates contain \[\ce{1 -> 4β}\] glycosidic linkage, EXCEPT ____________.
4-O-(α-D-Glucopyranosyl)-D-glucopyranose is ____________.
Which of the following monosaccharide is a ketohexose?
Which element among the following is not present in saccharine?
Which one of the following is generally applicable to polysaccharides?
Identify the number of secondary carbon atoms in glucose.
When 2 moles of stachyose is hydrolyzed, the number of moles of galactose obtained is ____________.
What is the molecular formula of glyceraldehyde?
How many moles of acetic acid are obtained in the reaction when one mole glucose is treated with excess acetic anhydride?
What is the product obtained when Br2 water reacts with glucose?
Which one of the following rotates the plane polarized light towards left?
Complete hydrolysis of cellulose gives ____________.
Glucose is an aldose. Which one of the following reactions is not expected with glucose?
Which of the following are epimers?
What are reducing and non-reducing sugars?
Is the following sugar, D-sugar or L-sugar?

Lactose is made of ______.
A molecule of stachyose contains how many carbon atoms?
Which carbon atoms of fructose are bonded together through oxygen forming fructofuranose?
Identify the monosaccharide containing only one asymmetric carbon atom in its molecule.
Starch and cellulose are compounds made up of many units of ______.
Which one is a non-reducing commercial sugar?
Consider the following reaction
\[\ce{A <-[Br2 - H2O] Glucose ->[HNO3] B}\]
Here, 'A' and 'B' are respectively.
\[\ce{CH2OH - CO - (CHOH)4 - CH2OH}\] is an example of ______.
Why carbohydrates are generally optically active.
Assign D/L configuration to the following monosaccharides:
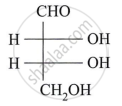
Describe the action of the following reagent on glucose:
dil. Nitric acid.
Carbohydrates that do not undergo hydrolysis further are called ______.
Write Fischer projection formulae for glucose.
CH2 OH - CO - (CHOH)4 - CH2 OH is an example of ______.
Why carbohydrates are generally optically active.
Why are carbohydrates generally optically active?
Why carbohydrates are generally optically active?
\[\ce{CH2OH - CO - (CHOH)4 - CH2 OH}\] is an example of ______.
\[\ce{CH2OH-CO-(CHOH)4-CH2OH}\] is an example of ______.
Two monosaccharides are held together by ______ bond.
Which of the following is used to separate glucose and fructose from hydrolysate of sucrose?
