Advertisements
Advertisements
प्रश्न
What is the action of the following reagents on glucose?
hydroxylamine
Advertisements
उत्तर
Glucose forms oxime by reaction with hydroxylamine:
\[\begin{array}{cc}
\phantom{}\ce{CHO}\phantom{..............}\ce{CH = N - OH}\phantom{..}\\
\phantom{.}|\phantom{..................}|\phantom{...............}\\
\ce{(CHOH)4 ->[NH2 - OH](CHOH)4 + H2O}\\
\phantom{}|\phantom{..................}|\phantom{..............}\\
\phantom{}\ce{\underset{\text{Glucose}}{CH2OH} \phantom{..........}\underset{\text{Oxime}}{CH2OH}}\phantom{........}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
\[\ce{CH2OH-CO-(CHOH)4-CH2OH}\] is an example of ______.
Give scientific reasons:
Hydrolysis of sucrose is called inversion.
Give two evidences for presence of formyl group in glucose.
Write the name of the polysaccharide used for the commercial preparation of glucose.
Write chemical reaction for following conversions
glucose into glucoxime
Explain the classification of carbohydrates with examples.
Which of the following type of carbohydrates cannot be hydrolyzed?
From the following identify an example of disaccharides.
From the following identify the group that is exclusively consists of polysaccharides.
Identify a non-reducing carbohydrate from the following.
Chitin is a/an ______.
Monosaccharides are ______ in nature.
Identify the CORRECT combination.
Which among the following type of linkages is present in cellulose?
Prolonged heating of glucose with hot HI results in the formation of ____________.
4-O-(α-D-Glucopyranosyl)-D-glucopyranose is ____________.
One mole of a carbohydrate on hydrolysis gives one mole of glucose and one mole of galactose. Identify the carbohydrate.
Which one of the following is an oligosaccharide?
The general formula for polysaccharide is ____________.
Stachyose is ____________.
Which one of the following is NOT soluble in water?
Glucose and gluconic acid treated with dilute nitric acid forms saccharic acid. What does this indicate?
Which of the following molecules reduces Fehling's solution?
Which among the following statements is true for amylose?
What is the number of hydroxyl groups present in lactic acid?
When 2 moles of stachyose is hydrolyzed, the number of moles of galactose obtained is ____________.
Which among the following reagents is used to confirm the presence of carbonyl group in glucose?
Which among the following sugars does not reduce Tollen's reagent?
What is the molecular formula of glyceraldehyde?
Which among the following is a product of hydrolysis of one mole raffinose?
What is the product obtained when Br2 water reacts with glucose?
Which one given below is a non-reducing sugar?
Assertion: A solution of sucrose in water is dextrorotatory. But on hydrolysis in the presence of little hydrochloric acid, it becomes levorotatory.
Reason: Sucrose hydrolysis gives equal amounts of glucose and fructose. As a result of this change in sign of rotation is observed.
Complete hydrolysis of cellulose gives ____________.
What are reducing and non-reducing sugars?
Why carbohydrates are generally optically active?
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Fructose
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Sucrose
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Lactose
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Maltose
The molecule of glucose is also called ______.
Which carbon atoms of fructose are bonded together through oxygen forming fructofuranose?
Match the Column I with Column II and choose the correct answer from options below:
| Column I | Column II |
| A. Purine | 1. Glycogen |
| B. Pyrimidine | 2. Cellulose |
| C. Structural polysaccharide | 3. Glucagon |
| D. Storage polysaccharide | 4. Adenine |
| 5. Cytosine |
Corn is immersed in boiling water. It is then cooled, and the solution becomes sweet. It is due to ______.
\[\ce{CH2OH-CO-(CHOH)4-CH2OH}\] is an example of ______.
Why carbohydrates are generally optically active.
Assign D/L configuration to the following monosaccharides:
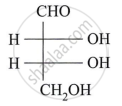
Carbohydrates that do not undergo hydrolysis further are called ______.
Write Fischer projection formulae for glucose.
Explain the hydrolysis of sucrose.
CH2 OH - CO - (CHOH)4 - CH2 OH is an example of ______.
Why carbohydrates are generally optically active.
\[\ce{CH2OH-CO-(CHOH)4-CH2OH}\] is an example of ______.
Why carbohydrates are generally optically active?
What are the number of chiral carbon atoms and the number of formyl groups, respectively, present in ribose?
