Advertisements
Advertisements
प्रश्न
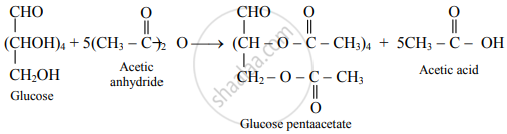
What is the action of the following reagents on glucose?
acetic anhydride
Advertisements
उत्तर
Glucose reacts with acetic anhydride to form glucose pentaacetate.
APPEARS IN
संबंधित प्रश्न
Give scientific reasons:
The disaccharide sucrose gives negative Tollens test while the disaccharide maltose gives a positive Tollens test.
Give scientific reasons:
Hydrolysis of sucrose is called inversion.
Draw a neat diagram for the following:
Haworth formula of maltose
The glycosidic linkage in maltose is formed between _______________
Write the name of the polysaccharide used for the commercial preparation of glucose.
Write chemical reaction for following conversions
glucose into glucoxime
Write chemical reaction for following conversions
glucose into gluconic acid
Classify the following carbohydrates into monosaccharide, disaccharide, oligosaccharide and polysaccharide:
glucose, cellulose, maltose, stachyose
Draw the Haworth projection structure of the following.
α-D-(–)-Fructofuranose
Write a chemical reaction to convert glucose into glucose cyanohydrin.
Write a commercial method for preparation of glucose.
By which of the following process formation of glycosidic bond occurs?
Which of the following type of carbohydrates cannot be hydrolyzed?
Identify the bond that links the carbon of two adjacent monosaccharides.
Which of the following is the basic unit of carbohydrates?
Carbohydrates can contain which of the following chemical groups?
From the following which is a heteropolysaccharide?
From the following identify the materials that are made up of cellulose.
i. Plant cell wall
ii. Exoskeleton of arthropods
iii. Paper from plant pulp
iv. Cotton fibre
Which is the product obtained, when Br2 water reacts with glucose?
Chitin is a/an ______.
The number of asymmetric carbon atoms in the glucose molecule is ____________.
Monosaccharides are ______ in nature.
Raffinose, sucrose and stachyose are respectively ____________.
4-O-(α-D-Glucopyranosyl)-D-glucopyranose is ____________.
One mole of a carbohydrate on hydrolysis gives one mole of glucose and one mole of galactose. Identify the carbohydrate.
Which element among the following is not present in saccharine?
The general formula for polysaccharide is ____________.
Which one of the following is NOT soluble in water?
Identify the number of secondary carbon atoms in glucose.
Identify the number of oxygen atoms present in saccharic acid?
What is the molecular formula of glyceraldehyde?
Identify the product Y in the following reaction.
\[\ce{Maltose ->[Hydrolysis] X ->[dil. HNO3][(excess)] Y}\]
How many moles of acetic acid are obtained in the reaction when one mole glucose is treated with excess acetic anhydride?
How many hydroxyl groups are present in Erythrulose?
Which one of the following rotates the plane polarized light towards left?
Which one given below is a non-reducing sugar?
Complete hydrolysis of cellulose gives ____________.
Which of the following are epimers?
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Sucrose
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Lactose
If 'n' represents total number of asymmetric carbon atoms in a compound, then the possible· number of optical isomers of the compound is ______.
Lactose is made of ______.
Which carbon atoms of fructose are bonded together through oxygen forming fructofuranose?
Starch and cellulose are compounds made up of many units of ______.
Why carbohydrates are generally optically active?
Carbohydrates that do not undergo hydrolysis further are called ______.
Why carbohydrates are generally optically active?
\[\ce{CH2OH - CO - (CHOH)4 - CH2 OH}\] is an example of ______.
\[\ce{CH2OH-CO-(CHOH)4-CH2OH}\] is an example of ______.
Why carbohydrates are generally optically active?
\[\ce{CH2OH - CO - (CHOH)4 - CH2OH}\] is an example of ______.
Why carbohydrates are generally optically active.
Why carbohydrates are generally optically active.
Two monosaccharides are held together by ______ bond.
What are the number of chiral carbon atoms and the number of formyl groups, respectively, present in ribose?
