Advertisements
Advertisements
प्रश्न
Draw the structure of α-D glucopyranose.
Advertisements
उत्तर

संबंधित प्रश्न
\[\ce{CH2OH-CO-(CHOH)4-CH2OH}\] is an example of ______.
Give scientific reasons:
The disaccharide sucrose gives negative Tollens test while the disaccharide maltose gives a positive Tollens test.
Draw a neat diagram for the following:
Haworth formula of maltose
Write chemical reaction for following conversions
glucose into glucoxime
Write chemical reaction for following conversions
glucose into gluconic acid
What is the action of the following reagents on glucose?
hydroxylamine
Define carbohydrates.
Draw the Haworth projection structure of the following.
α-D-(–)-Fructofuranose
Explain D and L configuration in sugars.
Which of the following is the basic unit of carbohydrates?
From the following identify the group that is exclusively consists of polysaccharides.
From the following which is a heteropolysaccharide?
Identify the WRONG statement.
Which is the product obtained, when Br2 water reacts with glucose?
The number of asymmetric carbon atoms in the glucose molecule is ____________.
From the following identify the two types of glucose polymers present in starch.
Match the columns and select the correct option.
| Column I | Column II | ||
| i. | Starch | a. | Animal storage molecule |
| ii. | Cellulose | b. | Plant storage molecule |
| iii. | Glycogen | c. | Heparin |
| iv. | Heteropolysaccharide | d. | Plant cell wall component |
Which reagent among the following is used to confirm presence of aldehydic carbonyl group in glucose?
Raffinose, sucrose and stachyose are respectively ____________.
Prolonged heating of glucose with hot HI results in the formation of ____________.
4-O-(α-D-Glucopyranosyl)-D-glucopyranose is ____________.
One mole of a carbohydrate on hydrolysis gives one mole of glucose and one mole of galactose. Identify the carbohydrate.
Which of the following monosaccharide is a ketohexose?
Which one of the following is an oligosaccharide?
\[\ce{CH3 - CH2 - CH2 - Br ->[Na][dry ether] A ->[Cl2] B}\]
Identify the product B in the above reaction.
Which of the following molecules reduces Fehling's solution?
Which following reagent is used to detect presence of five hydroxyl groups in a glucose molecule?
What is the number of hydroxyl groups present in lactic acid?
Identify the number of secondary carbon atoms in glucose.
On hydrolysis sucrose gives ____________.
When one mole of lactose is hydrolysed, the hydrolysate contains ____________.
\[\ce{Glucose ->[(HCN)] Product ->[(hydrolysis)] Product ->[(HI + Heat)] A}\], the compound A is:
Complete hydrolysis of cellulose gives ____________.
Which of the following are epimers?
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Sucrose
Write the structure of α-D (+) glucopyranose.
If 'n' represents total number of asymmetric carbon atoms in a compound, then the possible· number of optical isomers of the compound is ______.
Glucose on oxidation with bromine water yields gluconic acid. This reaction confirms the presence of ______.
Which one is a non-reducing commercial sugar?
Formation of gluconic acid from glucose by oxidation using Br2 water.
The glycosidic linkage present in maltose is ______.
\[\ce{CH2OH-CO-(CHOH)4-CH2OH}\] is an example of ______.
Assign D/L configuration to the following monosaccharides:
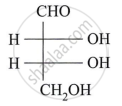
Describe the action of the following reagent on glucose:
Bromine water
Write Fischer projection formulae for glucose.
Write the ring structure of glucose.
Why carbohydrates are generally optically active.
Why carbohydrates are generally optically active.
Why carbohydrates are generally optically active.
