Advertisements
Advertisements
प्रश्न
Draw the structure of α-D glucopyranose.
Advertisements
उत्तर

संबंधित प्रश्न
Give scientific reasons:
The disaccharide sucrose gives negative Tollens test while the disaccharide maltose gives a positive Tollens test.
Explain the preparation of glucose from sucrose.
Classify the following carbohydrates into monosaccharide, disaccharide, oligosaccharide and polysaccharide:
glucose, cellulose, maltose, stachyose
Explain the classification of carbohydrates with examples.
What is the action of the following reagents on glucose?
hydrogen iodide
Explain D and L configuration in sugars.
By which of the following process formation of glycosidic bond occurs?
Which enzymes would work sequentially on potatoes consumed by an individual?
Formation of lactose by conden ation results in the release of how many molecules of water?
______ is the prosthetic group of glycoproteins.
Fructose is the fruit sugar and chemically it is ketohexose but it has a ______ rather than a ______.
Identify the WRONG statement.
Identify a non-reducing carbohydrate from the following.
Which is the product obtained, when Br2 water reacts with glucose?
The number of asymmetric carbon atoms in the glucose molecule is ____________.
Identify the CORRECT combination.
Which among the following type of linkages is present in cellulose?
Raffinose, sucrose and stachyose are respectively ____________.
Prolonged heating of glucose with hot HI results in the formation of ____________.
4-O-(α-D-Glucopyranosyl)-D-glucopyranose is ____________.
One mole of a carbohydrate on hydrolysis gives one mole of glucose and one mole of galactose. Identify the carbohydrate.
Stachyose is ____________.
Which of the following statements is NOT true for glyceraldehyde?
Which of the following molecules reduces Fehling's solution?
Which among the following sugars does not reduce Tollen's reagent?
Identify the number of oxygen atoms present in saccharic acid?
What is the molecular formula of glyceraldehyde?
How many moles of acetic acid are obtained in the reaction when one mole glucose is treated with excess acetic anhydride?
Which among the following is a product of hydrolysis of one mole raffinose?
How many hydroxyl groups are present in Erythrulose?
The number of sp2 and sp3 hybridised carbon in fructose are respectively ____________.
Complete hydrolysis of cellulose gives ____________.
α-D (+) Glucose and β-D (+) glucose are ____________.
Which of the following are epimers?
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Starch
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Sucrose
The molecule of glucose is also called ______.
Match the Column I with Column II and choose the correct answer from options below:
| Column I | Column II |
| A. Purine | 1. Glycogen |
| B. Pyrimidine | 2. Cellulose |
| C. Structural polysaccharide | 3. Glucagon |
| D. Storage polysaccharide | 4. Adenine |
| 5. Cytosine |
Identify the monosaccharide containing only one asymmetric carbon atom in its molecule.
Corn is immersed in boiling water. It is then cooled, and the solution becomes sweet. It is due to ______.
Consider the following reaction
\[\ce{A <-[Br2 - H2O] Glucose ->[HNO3] B}\]
Here, 'A' and 'B' are respectively.
Identify the product obtained in the following conversion.
\[\ce{Glucose ->[(O)][Br2 water] Product}\]
The glycosidic linkage present in maltose is ______.
\[\ce{CH2OH-CO-(CHOH)4-CH2OH}\] is an example of ______.
Why carbohydrates are generally optically active?
Assign D/L configuration to the following monosaccharides:
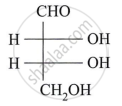
Describe the action of the following reagent on glucose:
Bromine water
Write the ring structure of glucose.
Why carbohydrates are generally optically active.
CH2 OH - CO - (CHOH)4 - CH2 OH is an example of ______.
Why carbohydrates are generally optically active?
\[\ce{CH2OH - CO - (CHOH)4 - CH2OH}\] is an example of ______.
Why carbohydrates are generally optically active.
