Advertisements
Advertisements
प्रश्न
Write the name of the polysaccharide used for the commercial preparation of glucose.
Advertisements
उत्तर
Starch
APPEARS IN
संबंधित प्रश्न
Draw a neat diagram for the following:
Haworth formula of maltose
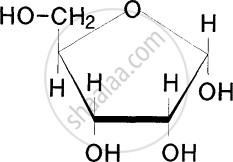
Draw the structure of α-D glucopyranose.
Write the name of the unit to which glucose unit is linked to from sucrose.
Explain the preparation of glucose from sucrose.
Write chemical reaction for following conversions
glucose into gluconic acid
What is the action of the following reagents on glucose?
hydrogen iodide
Define carbohydrates.
Draw the Haworth projection structure of the following.
α-D-(–)-Fructofuranose
Explain D and L configuration in sugars.
Write a chemical reaction to convert glucose into glucose cyanohydrin.
Write a commercial method for preparation of glucose.
Identify the given structure 'P' and 'Q'.

Which enzymes would work sequentially on potatoes consumed by an individual?
Which of the following carbohydrate is sweet to taste?
Carbohydrates can contain which of the following chemical groups?
From the following which is a heteropolysaccharide?
______ is the prosthetic group of glycoproteins.
Identify the sugar having the molecular formula C6H1206.
Identify the WRONG statement.
Chitin is a/an ______.
Which of the following monosaccharide is a ketohexose?
Which one of the following is an oligosaccharide?
Which one of the following is a carbohydrate but does NOT follow the general formula of carbohydrate?
Which one of the following is Tetrose sugar?
Which one of the following is NOT soluble in water?
On hydrolysis sucrose gives ____________.
What is the quantity of glucose obtained when 68.4 g of sucrose is hydrolyzed in laboratory under ideal condition?
(Molar mass of sucrose = 342 g mol−1)
Identify the number of oxygen atoms present in saccharic acid?
When one mole of lactose is hydrolysed, the hydrolysate contains ____________.
Which among the following observations suggests that glucose also exists in cyclic form?
How many optical isomers are possible for a compound having four asymmetric carbon atoms?
Which one of the following rotates the plane polarized light towards left?
Assertion: A solution of sucrose in water is dextrorotatory. But on hydrolysis in the presence of little hydrochloric acid, it becomes levorotatory.
Reason: Sucrose hydrolysis gives equal amounts of glucose and fructose. As a result of this change in sign of rotation is observed.
Complete hydrolysis of cellulose gives ____________.
α-D (+) Glucose and β-D (+) glucose are ____________.
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Fructose
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Sucrose
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Lactose
The molecule of glucose is also called ______.
If 'n' represents total number of asymmetric carbon atoms in a compound, then the possible· number of optical isomers of the compound is ______.
A molecule of stachyose contains how many carbon atoms?
Starch and cellulose are compounds made up of many units of ______.
The glycosidic linkage present in maltose is ______.
\[\ce{CH2OH - CO - (CHOH)4 - CH2OH}\] is an example of ______.
Why carbohydrates are generally optically active.
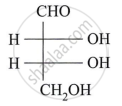
Assign D/L configuration to the following monosaccharides:
Describe the action of the following reagent on glucose:
Bromine water
Describe the action of the following reagent on glucose:
dil. Nitric acid.
Write the ring structure of glucose.
CH2 OH - CO - (CHOH)4 - CH2 OH is an example of ______.
The linkage present in Lactose is ______.
Why carbohydrates are generally optically active?
Why carbohydrates are generally optically active?
Why carbohydrates are generally optically active.
Two monosaccharides are held together by ______ bond.
Which of the following is used to separate glucose and fructose from hydrolysate of sucrose?
