Advertisements
Advertisements
प्रश्न
Write the name of the polysaccharide used for the commercial preparation of glucose.
Advertisements
उत्तर
Starch
APPEARS IN
संबंधित प्रश्न
Glucose on oxidation with dilute nitric acid gives _______________
The glycosidic linkage in maltose is formed between _______________
Write chemical reaction for following conversions
glucose into glucoxime
Explain the classification of carbohydrates with examples.
What is the action of the following reagents on glucose?
acetic anhydride
What is the action of the following reagents on glucose?
hydroxylamine
Draw the Haworth projection structure of the following.
α-D-(–)-Fructofuranose
By which of the following process formation of glycosidic bond occurs?
Formation of lactose by conden ation results in the release of how many molecules of water?
Carbohydrates can contain which of the following chemical groups?
Fructose is the fruit sugar and chemically it is ketohexose but it has a ______ rather than a ______.
From the following identify the materials that are made up of cellulose.
i. Plant cell wall
ii. Exoskeleton of arthropods
iii. Paper from plant pulp
iv. Cotton fibre
Which among the following compounds is obtained when glucose reacts with hydrogen cyanide?
Which is the product obtained, when Br2 water reacts with glucose?
The number of asymmetric carbon atoms in the glucose molecule is ____________.
Monosaccharides are ______ in nature.
Which among the following type of linkages is present in cellulose?
The general formula for polysaccharide is ____________.
Which following reagent is used to detect presence of five hydroxyl groups in a glucose molecule?
Which among the following statements is true for amylose?
Which among the following reagents is used to confirm the presence of carbonyl group in glucose?
Which among the following sugars does not reduce Tollen's reagent?
What is the quantity of glucose obtained when 68.4 g of sucrose is hydrolyzed in laboratory under ideal condition?
(Molar mass of sucrose = 342 g mol−1)
Identify the product Y in the following reaction.
\[\ce{Maltose ->[Hydrolysis] X ->[dil. HNO3][(excess)] Y}\]
How many hydroxyl groups are present in Erythrulose?
How many optical isomers are possible for a compound having four asymmetric carbon atoms?
Which one of the following rotates the plane polarized light towards left?
The correct corresponding order of names of four aldoses with configuration given below Respectively is:
Assertion: A solution of sucrose in water is dextrorotatory. But on hydrolysis in the presence of little hydrochloric acid, it becomes levorotatory.
Reason: Sucrose hydrolysis gives equal amounts of glucose and fructose. As a result of this change in sign of rotation is observed.
Complete hydrolysis of cellulose gives ____________.
Glucose is an aldose. Which one of the following reactions is not expected with glucose?
Which of the following are epimers?
Why carbohydrates are generally optically active?
Is the following sugar, D-sugar or L-sugar?

Which of the following groups contain polysaccharides?
Glucose on oxidation with bromine water yields gluconic acid. This reaction confirms the presence of ______.
Which carbon atoms of fructose are bonded together through oxygen forming fructofuranose?
The glycosidic linkage present in maltose is ______.
\[\ce{CH2OH - CO - (CHOH)4 - CH2OH}\] is an example of ______.
\[\ce{CH2OH-CO-(CHOH)4-CH2OH}\] is an example of ______.
Why carbohydrates are generally optically active.
Assign D/L configuration to the following monosaccharides:
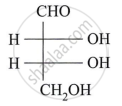
Describe the action of the following reagent on glucose:
dil. Nitric acid.
Write the ring structure of glucose.
Why carbohydrates are generally optically active.
The linkage present in Lactose is ______.
What are the number of chiral carbon atoms and the number of formyl groups, respectively, present in ribose?
The sugar found in milk is ______.
