Advertisements
Advertisements
प्रश्न
Define carbohydrates.
Advertisements
उत्तर
Carbohydrates are optically active polyhydroxy aldehydes or polyhydroxy ketones or compounds that can be hydrolysed to polyhydroxy aldehydes or polyhydroxy ketones.
APPEARS IN
संबंधित प्रश्न
Draw a neat diagram for the following:
Haworth formula of glucopyranose.
Write the name of the polysaccharide used for the commercial preparation of glucose.
Write the name of the unit to which glucose unit is linked to from sucrose.
Explain the preparation of glucose from sucrose.
Write chemical reaction for following conversions
glucose into gluconic acid
Draw the Haworth projection structure of the following.
α-D-(–)-Fructofuranose
What is monosaccharide?
By which of the following process formation of glycosidic bond occurs?
Which of the following type of carbohydrates cannot be hydrolyzed?
Identify the given structure 'P' and 'Q'.

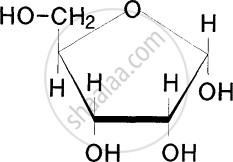
Which of the following monosaccharides is a tetrose sugar?
The reserve food material in animals is ____________.
Fructose is the fruit sugar and chemically it is ketohexose but it has a ______ rather than a ______.
Identify the WRONG statement.
Identify a non-reducing carbohydrate from the following.
Which is the product obtained, when Br2 water reacts with glucose?
Match the Column I and Column II select the correct option.
| Column I | Column II | ||
| i. | Monosaccharides | a. | Stored energy in animals |
| ii. | Glycogen | b. | Polymer made from ß-glucose molecules |
| iii. | Cellulose | c. | Exoskeleton of arthropods |
| iv. | Chitin | d. | Building blocks of carbohydrates |
| e. | Plant hormone |
Monosaccharides are ______ in nature.
Which reagent among the following is used to confirm presence of aldehydic carbonyl group in glucose?
Prolonged heating of glucose with hot HI results in the formation of ____________.
4-O-(α-D-Glucopyranosyl)-D-glucopyranose is ____________.
Identify the INCORRECT statement regarding glucose.
Which of the following monosaccharide is a ketohexose?
\[\ce{CH3 - CH2 - CH2 - Br ->[Na][dry ether] A ->[Cl2] B}\]
Identify the product B in the above reaction.
Which following reagent is used to detect presence of five hydroxyl groups in a glucose molecule?
Which among the following statements is true for amylose?
Which one of the following sugar does NOT have same empirical fonnula as that of carbohydrate?
Which of the following statement is NOT true about saccharic acid?
Which among the following sugars does not reduce Tollen's reagent?
Identify the number of oxygen atoms present in saccharic acid?
Which among the following reagents is used for conversion of glucose to glucoxime?
Which among the following reagents is used to obtain gluconic acid from glucose?
Which among the following is a product of hydrolysis of one mole raffinose?
When one mole of lactose is hydrolysed, the hydrolysate contains ____________.
The correct corresponding order of names of four aldoses with configuration given below Respectively is:
α-D (+) Glucose and β-D (+) glucose are ____________.
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Starch
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Fructose
The two monosaccharides in a disaccharide are held together by ______ bonds.
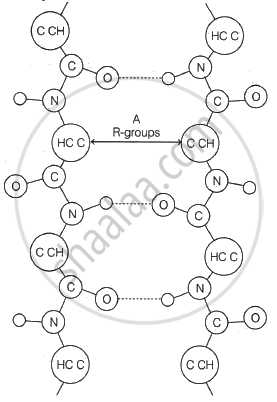
Identify the monosaccharide containing only one asymmetric carbon atom in its molecule.
Which one is a non-reducing commercial sugar?
The glycosidic linkage present in maltose is ______.
Why carbohydrates are generally optically active.
Why carbohydrates are generally optically active.
Write Fischer projection formulae for glucose.
CH2 OH - CO - (CHOH)4 - CH2 OH is an example of ______.
Why carbohydrates are generally optically active.
The linkage present in Lactose is ______.
Why carbohydrates are generally optically active?
\[\ce{CH2OH - CO - (CHOH)4 - CH2OH}\] is an example of ______.
Why carbohydrates are generally optically active.
